Cdh1-anaphase-promoting complex targets Skp2 for destruction in transforming growth factor beta-induced growth inhibition
- PMID: 17283060
- PMCID: PMC1899917
- DOI: 10.1128/MCB.01830-06
Cdh1-anaphase-promoting complex targets Skp2 for destruction in transforming growth factor beta-induced growth inhibition
Abstract
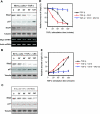
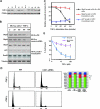
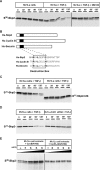
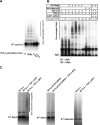
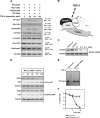
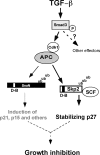
As a subunit of a ubiquitin ligase, Skp2 is implicated in facilitating cell cycle progression via degradation of various protein targets. We report here that Skp2 is rapidly degraded following cellular stimulation by the cytokine transforming growth factor beta (TGF-beta) and that this degradation stabilizes the cell cycle arrest protein p27. The Skp2 degradation is mediated by Cdh1-anaphase-promoting complex (APC), as shown by depletion of Cdh1 with small interfering RNA, and by reconstitution of ubiquitylation reactions in a purified system. Blockage of Skp2 degradation greatly reduces TGF-beta-induced cell cycle arrest, as does expression of a nondegradable Skp2 mutant. Furthermore, we demonstrate that TGF-beta-induced Skp2 degradation is mediated by the Smad cascade. The degradation of Skp2 stabilizes p27, thereby ensuring TGF-beta-induced cell cycle arrest. These results identify a novel mechanism for tumor suppression by TGF-beta and explain why dysfunction of APC in the TGF-beta pathway in responsive cells is associated with cancer.
Figures

Similar articles
-
TGF-β activates APC through Cdh1 binding for Cks1 and Skp2 proteasomal destruction stabilizing p27kip1 for normal endometrial growth.Cell Cycle. 2016;15(7):931-47. doi: 10.1080/15384101.2016.1150393. Cell Cycle. 2016. PMID: 26963853 Free PMC article.
-
Control of the SCF(Skp2-Cks1) ubiquitin ligase by the APC/C(Cdh1) ubiquitin ligase.Nature. 2004 Mar 11;428(6979):190-3. doi: 10.1038/nature02330. Nature. 2004. PMID: 15014502
-
Cell adhesion induces p27Kip1-associated cell-cycle arrest through down-regulation of the SCFSkp2 ubiquitin ligase pathway in mantle-cell and other non-Hodgkin B-cell lymphomas.Blood. 2007 Sep 1;110(5):1631-8. doi: 10.1182/blood-2006-11-060350. Epub 2007 May 14. Blood. 2007. PMID: 17502456 Free PMC article. Clinical Trial.
-
Don't skip the G1 phase: how APC/CCdh1 keeps SCFSKP2 in check.Cell Cycle. 2004 Jul;3(7):850-2. Epub 2004 Jul 14. Cell Cycle. 2004. PMID: 15190201 Review.
-
Methods to measure ubiquitin-dependent proteolysis mediated by the anaphase-promoting complex.Methods. 2006 Jan;38(1):39-51. doi: 10.1016/j.ymeth.2005.07.005. Methods. 2006. PMID: 16343932 Review.
Cited by
-
Regulation of the anaphase-promoting complex-separase cascade by transforming growth factor-beta modulates mitotic progression in bone marrow stromal cells.Mol Biol Cell. 2008 Dec;19(12):5446-55. doi: 10.1091/mbc.e08-03-0289. Epub 2008 Oct 8. Mol Biol Cell. 2008. PMID: 18843049 Free PMC article.
-
Novel insight into KLF4 proteolytic regulation in estrogen receptor signaling and breast carcinogenesis.J Biol Chem. 2012 Apr 20;287(17):13584-97. doi: 10.1074/jbc.M112.343566. Epub 2012 Mar 2. J Biol Chem. 2012. PMID: 22389506 Free PMC article.
-
Depletion of DNA damage binding protein 2 sensitizes triple-negative breast cancer cells to poly ADP-ribose polymerase inhibition by destabilizing Rad51.Cancer Sci. 2019 Nov;110(11):3543-3552. doi: 10.1111/cas.14201. Epub 2019 Oct 6. Cancer Sci. 2019. PMID: 31541611 Free PMC article.
-
14-3-3ζ targeting induced senescence in Hep-2 laryngeal cancer cell through deneddylation of Cullin1 in the Skp1-Cullin-F-box protein complex.Cell Prolif. 2019 Sep;52(5):e12654. doi: 10.1111/cpr.12654. Epub 2019 Jun 21. Cell Prolif. 2019. PMID: 31222857 Free PMC article.
-
Hypoxia and cell cycle regulation of the von Hippel-Lindau tumor suppressor.Oncogene. 2011 Jan 6;30(1):21-31. doi: 10.1038/onc.2010.395. Epub 2010 Aug 30. Oncogene. 2011. PMID: 20802534 Free PMC article.
References
-
- Ang, X. L., and J. W. Harper. 2004. Interwoven ubiquitination oscillators and control of cell cycle transitions. Sci. STKE 2004:e31. - PubMed
-
- Bashir, T., N. V. Dorrello, V. Amador, D. Guardavaccaro, and M. Pagano. 2004. Control of the SCF (Skp2-Cks1) ubiquitin ligase by the APC/C (Cdh1) ubiquitin ligase. Nature 428:190-193. - PubMed
-
- Carrano, A. C., E. Eytan, A. Hershko, and M. Pagano. 1999. SKP2 is required for ubiquitin-mediated degradation of the CDK inhibitor p27. Nat. Cell Biol. 1:193-199. - PubMed
-
- Catzavelos, C., N. Bhattacharya, Y. C. Ung, J. A. Wilson, L. Roncari, C. Sandhu, P. Shaw, H. Yeger, I. Morava-Protzner, L. Kapusta, E. Franssen, K. I. Pritchard, and J. M. Slingerland. 1997. Decreased levels of the cell-cycle inhibitor p27Kip1 protein: prognostic implications in primary breast cancer. Nat. Med. 3:227-230. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
