Involvement of dihydroceramide desaturase in cell cycle progression in human neuroblastoma cells
- PMID: 17283068
- PMCID: PMC2084375
- DOI: 10.1074/jbc.M700647200
Involvement of dihydroceramide desaturase in cell cycle progression in human neuroblastoma cells
Abstract
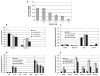
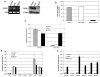
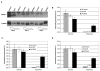
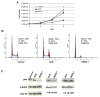
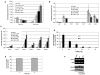
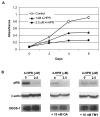
The role of dihydroceramide desaturase as a key enzyme in the de novo pathway of ceramide generation was investigated in human neuroblastoma cells (SMS-KCNR). A novel assay using water-soluble analogs of dihydroceramide, dihydroceramidoids (D-erythro-dhCCPS analogs), was used to measure desaturase activity in situ. Conversion of D-erythro-2-N-[12'-(1''-pyridinium)-dodecanoyl]-4,5-dihydrosphingosine bromide (C(12)-dhCCPS) to its 4,5-desaturated counterpart, D-erythro-2-N-[12'-(1''-pyridinium)dodecanoyl]sphingosine bromide (C(12)-CCPS), was determined by liquid chromatography/mass spectrometry analysis. The validity of the assay was confirmed using C(8)-cyclopropenylceramide, a competitive inhibitor of dihydroceramide desaturase. A human homolog (DEGS-1) of the Drosophila melanogaster des-1 gene was recently identified and reported to have desaturase activity. Transfection of SMS-KCNR cells with small interfering RNA to DEGS-1 significantly blocked the conversion of C(12)-dhCCPS to C(12)-CCPS. The associated accumulation of endogenous dihydroceramides confirmed DEGS-1 as the main active dihydroceramide desaturase in these cells. The partial loss of DEGS-1 inhibited cell growth, with cell cycle arrest at G(0)/G(1). This was accompanied by a significant decrease in the amount of phosphorylated retinoblastoma protein. This hypophosphorylation was inhibited by tautomycin and not by okadaic acid, suggesting the involvement of protein phosphatase 1. Additionally, we found that treatment of SMS-KCNR cells with fenretinide inhibited desaturase activity in a dose-dependent manner. An increase in dihydroceramides (but not ceramides) paralleled this process as measured by liquid chromatography/mass spectrometry. There were no effects on the mRNA or protein levels of DEGS-1, suggesting that fenretinide acts at the post-translational level as an inhibitor of this enzyme. Tautomycin was also able to block the hypophosphorylation of the retinoblastoma protein observed upon fenretinide treatment. These findings suggest a novel biological function for dihydroceramides.
Figures

References
-
- Hannun YA, Obeid LM. J Biol Chem. 2002;277(29):25847–25850. - PubMed
-
- Hannun YA. Adv Exp Med Biol. 1997;400A:305–312. - PubMed
-
- Hannun YA, Luberto C. Trends Cell Biol. 2000;10(2):73–80. - PubMed
-
- Hannun YA, Obeid LM. Biochem Soc Trans. 1997;25(4):1171–1175. - PubMed
-
- Ogretmen B, Hannun YA. Nat Rev Cancer. 2004;4(8):604–616. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

