Death-effector domain-containing protein DEDD is an inhibitor of mitotic Cdk1/cyclin B1
- PMID: 17283331
- PMCID: PMC1793902
- DOI: 10.1073/pnas.0611167104
Death-effector domain-containing protein DEDD is an inhibitor of mitotic Cdk1/cyclin B1
Abstract
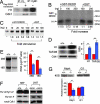
Accumulating evidence has shown that many molecules, including some cyclin-dependent kinases (Cdks) and cyclins, as well as the death-effector domain (DED)-containing FADD, function for both apoptosis and cell cycle. Here we identified that DEDD, which also possesses the DED domain, acts as a novel inhibitor of the mitotic Cdk1/cyclin B1 complex. DEDD associates with mitotic Cdk1/cyclin B1 complexes via direct binding to cyclin B1 and reduces their function. In agreement, kinase activity of nuclear Cdk1/cyclin B1 in DEDD-null (DEDD-/-) embryonic fibroblasts is increased compared with that in DEDD+/+ cells, which results in accelerated mitotic progression, thus exhibiting a shortened G2/M stage. Interestingly, DEDD-/- cells also demonstrated decreased G1 duration, which perhaps enhanced the overall reduction in rRNA amounts and cell volume, primarily caused by the rapid termination of rRNA synthesis before cell division. Likewise, DEDD-/- mice show decreased body and organ weights relative to DEDD+/+ mice. Thus, DEDD is an impeder of cell mitosis, and its absence critically influences cell and body size via modulation of rRNA synthesis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

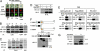

Similar articles
-
Two distinct controls of mitotic cdk1/cyclin B1 activity requisite for cell growth prior to cell division.Cell Cycle. 2007 Jun 15;6(12):1419-25. Epub 2007 May 7. Cell Cycle. 2007. PMID: 17592253 Review.
-
The death effector domain-containing DEDD supports S6K1 activity via preventing Cdk1-dependent inhibitory phosphorylation.J Biol Chem. 2009 Feb 20;284(8):5050-5. doi: 10.1074/jbc.M808598200. Epub 2008 Dec 22. J Biol Chem. 2009. PMID: 19106089 Free PMC article.
-
p21-Mediated nuclear retention of cyclin B1-Cdk1 in response to genotoxic stress.Mol Biol Cell. 2004 Sep;15(9):3965-76. doi: 10.1091/mbc.e03-12-0871. Epub 2004 Jun 4. Mol Biol Cell. 2004. PMID: 15181148 Free PMC article.
-
Mitotic phosphorylation of Aki1 at Ser208 by cyclin B1-Cdk1 complex.Biochem Biophys Res Commun. 2010 Mar 19;393(4):872-6. doi: 10.1016/j.bbrc.2010.02.103. Epub 2010 Feb 18. Biochem Biophys Res Commun. 2010. PMID: 20171170
-
Cyclin B1 and CDK1: nuclear localization and upstream regulators.Prog Cell Cycle Res. 2003;5:335-47. Prog Cell Cycle Res. 2003. PMID: 14593728 Review.
Cited by
-
Integrative analysis of 1q23.3 copy-number gain in metastatic urothelial carcinoma.Clin Cancer Res. 2014 Apr 1;20(7):1873-83. doi: 10.1158/1078-0432.CCR-13-0759. Epub 2014 Jan 31. Clin Cancer Res. 2014. PMID: 24486590 Free PMC article.
-
Developmental regulation of decidual cell polyploidy at the site of implantation.Front Biosci (Schol Ed). 2012 Jun 1;4(4):1475-86. doi: 10.2741/s347. Front Biosci (Schol Ed). 2012. PMID: 22652887 Free PMC article. Review.
-
Inside the Endometrial Cell Signaling Subway: Mind the Gap(s).Int J Mol Sci. 2018 Aug 21;19(9):2477. doi: 10.3390/ijms19092477. Int J Mol Sci. 2018. PMID: 30134622 Free PMC article. Review.
-
Genes associated with body weight gain and feed intake identified by meta-analysis of the mesenteric fat from crossbred beef steers.PLoS One. 2020 Jan 7;15(1):e0227154. doi: 10.1371/journal.pone.0227154. eCollection 2020. PLoS One. 2020. PMID: 31910243 Free PMC article.
-
Genetic Pathways of Neuroregeneration in a Novel Mild Traumatic Brain Injury Model in Adult Zebrafish.eNeuro. 2018 Jan 3;5(1):ENEURO.0208-17.2017. doi: 10.1523/ENEURO.0208-17.2017. eCollection 2018 Jan-Feb. eNeuro. 2018. PMID: 29302617 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

