Ectopic brown adipose tissue in muscle provides a mechanism for differences in risk of metabolic syndrome in mice
- PMID: 17283342
- PMCID: PMC1892979
- DOI: 10.1073/pnas.0610416104
Ectopic brown adipose tissue in muscle provides a mechanism for differences in risk of metabolic syndrome in mice
Abstract
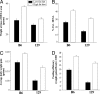
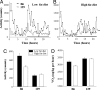
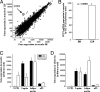
C57BL/6 (B6) mice subjected to a high-fat diet develop metabolic syndrome with obesity, hyperglycemia, and insulin resistance, whereas 129S6/SvEvTac (129) mice are relatively protected from this disorder because of differences in higher basal energy expenditure in 129 mice, leading to lower weight gain. At a molecular level, this difference correlates with a marked higher expression of uncoupling protein 1 (UCP1) and a higher degree of uncoupling in vitro in mitochondria isolated from muscle of 129 versus B6 mice. Detailed histological examination, however, reveals that this UCP1 is in mitochondria of brown adipocytes interspersed between muscle bundles. Indeed, the number of UCP1-positive brown fat cells in intermuscular fat in 129 mice is >700-fold higher than in B6 mice. These brown fat cells are subject to further up-regulation of UCP1 after stimulation with a beta3-adrenergic receptor agonist. Thus, ectopic deposits of brown adipose tissue in intermuscular depots with regulatable expression of UCP1 provide a genetically based mechanism of protection from weight gain and metabolic syndrome between strains of mice.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
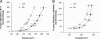


References
-
- Bogardus C. Diabetes Care. 1993;16:228–231. - PubMed
-
- Lee ET, Howard BV, Savage PJ, Cowan LD, Fabsitz RR, Oopik AJ, Yeh J, Go O, Robbins DC, Welty TK. Diabetes Care. 1995;18:599–610. - PubMed
-
- Meigs JB, Cupples LA, Wilson PW. Diabetes. 2000;49:2201–2207. - PubMed
-
- Almind K, Kulkarni RN, Lannon S, Kahn C. Diabetes. 2003;52:1535–1543. - PubMed
-
- Kulkarni RN, Almind K, Goren HJ, Winnay JN, Ueki K, Okada T, Kahn CR. Diabetes. 2003;52:1528–1534. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

