Effective inhibition of porcine transmissible gastroenteritis virus replication in ST cells by shRNAs targeting RNA-dependent RNA polymerase gene
- PMID: 17287033
- PMCID: PMC7114347
- DOI: 10.1016/j.antiviral.2006.12.007
Effective inhibition of porcine transmissible gastroenteritis virus replication in ST cells by shRNAs targeting RNA-dependent RNA polymerase gene
Abstract
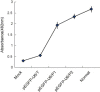
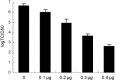
Transmissible gastroenteritis virus (TGEV) is identified as one of the most important pathogenic agents during swine enteric infection, leading to high mortality in neonatal pigs and severe annual economic loss in swine-producing areas. Up to date, various vaccines developed against TGEV still need to be improved. To exploit the possibility of using RNA interference (RNAi) as a strategy against TGEV infection, two shRNA-expressing plasmids (pEGFP-U6/P1 and pEGFP-U6/P2) targeting the RNA-dependent RNA polymerase (RdRp) gene of TGEV were constructed and transfected into swine testicular (ST) cells. The cytopathic effect (CPE) and MTS assays demonstrated that both shRNAs were capable of protecting cells against TGEV invasion with very high specificity and efficiency. A real-time quantitative RT-PCR further confirmed that the amounts of viral RNAs in cell cultures pre-transfected with the two plasmids were reduced by 95.2% and up to 100%, respectively. Our results suggest that RNAi might be a promising new strategy against TGEV infection.
Figures

Similar articles
-
In vitro inhibition of transmissible gastroenteritis coronavirus replication in swine testicular cells by short hairpin RNAs targeting the ORF 7 gene.Virol J. 2012 Aug 28;9:176. doi: 10.1186/1743-422X-9-176. Virol J. 2012. PMID: 22929207 Free PMC article.
-
Inhibition of porcine transmissible gastroenteritis virus infection in porcine kidney cells using short hairpin RNAs targeting the membrane gene.Virus Genes. 2017 Apr;53(2):226-232. doi: 10.1007/s11262-016-1409-8. Epub 2016 Nov 15. Virus Genes. 2017. PMID: 27848068 Free PMC article.
-
Inhibition of porcine transmissible gastroenteritis virus (TGEV) replication in mini-pigs by shRNA.Virus Res. 2010 Apr;149(1):51-5. doi: 10.1016/j.virusres.2009.12.012. Epub 2010 Jan 18. Virus Res. 2010. PMID: 20080134 Free PMC article.
-
An overview of immunological and genetic methods for detecting swine coronaviruses, transmissible gastroenteritis virus, and porcine respiratory coronavirus in tissues.Adv Exp Med Biol. 1997;412:37-46. doi: 10.1007/978-1-4899-1828-4_4. Adv Exp Med Biol. 1997. PMID: 9191988 Review.
-
Transmissible gastroenteritis virus infection: a vanishing specter.Dtsch Tierarztl Wochenschr. 2006 Apr;113(4):157-9. Dtsch Tierarztl Wochenschr. 2006. PMID: 16716052 Review.
Cited by
-
In vitro inhibition of transmissible gastroenteritis coronavirus replication in swine testicular cells by short hairpin RNAs targeting the ORF 7 gene.Virol J. 2012 Aug 28;9:176. doi: 10.1186/1743-422X-9-176. Virol J. 2012. PMID: 22929207 Free PMC article.
-
Identification and characterisation of the porcine 7SK RNA polymerase III promoter for short hairpin RNA expression.J RNAi Gene Silencing. 2008 May 27;4(1):289-94. J RNAi Gene Silencing. 2008. PMID: 19771238 Free PMC article.
-
All-Trans Retinoic Acid Attenuates Transmissible Gastroenteritis Virus-Induced Apoptosis in IPEC-J2 Cells via Inhibiting ROS-Mediated P38MAPK Signaling Pathway.Antioxidants (Basel). 2022 Feb 10;11(2):345. doi: 10.3390/antiox11020345. Antioxidants (Basel). 2022. PMID: 35204227 Free PMC article.
-
Inhibition of porcine transmissible gastroenteritis virus infection in porcine kidney cells using short hairpin RNAs targeting the membrane gene.Virus Genes. 2017 Apr;53(2):226-232. doi: 10.1007/s11262-016-1409-8. Epub 2016 Nov 15. Virus Genes. 2017. PMID: 27848068 Free PMC article.
-
RNA interference targeting nucleocapsid protein inhibits porcine reproductive and respiratory syndrome virus replication in Marc-145 cells.J Microbiol. 2014 Apr;52(4):333-9. doi: 10.1007/s12275-014-3419-3. Epub 2014 Mar 29. J Microbiol. 2014. PMID: 24682995 Free PMC article.
References
-
- Arenz C., Schepers U. RNA interference: from an ancient mechanism to a state of the art therapeutic application? Naturwissenschaften. 2003;90:345–359. - PubMed
-
- Bagasra O., Prilliman K.R. RNA interference: the molecular immune system. J. Mol. Histol. 2004;35:545–553. - PubMed
-
- Borkhardt A., Heidenreich O. RNA interference as a potential tool in the treatment of leukaemia. Exp. Opin. Biol. Ther. 2004;4:1921–1929. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

