Quantitative and mechanism-based investigation of post-nuclear delivery events between adenovirus and lipoplex
- PMID: 17287293
- PMCID: PMC1865055
- DOI: 10.1093/nar/gkl1165
Quantitative and mechanism-based investigation of post-nuclear delivery events between adenovirus and lipoplex
Abstract
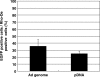
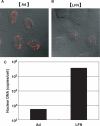
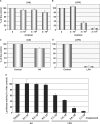
Quantitative and mechanism-based information on differences in transfection efficiency between viral and non-viral vectors would be highly useful for improving the effectiveness of non-viral vectors. A previous quantitative comparison of intracellular trafficking between adenovirus and LipofectAMINE PLUS (LFN) revealed that the three orders of magnitude lower transfection efficiency of LFN was dominantly rate limited by the post-nuclear delivery process. In the present study, the contribution of transcription and translation processes to the overall differences in the transgene expression efficiency of nucleus-delivered DNA was independently evaluated by quantifying mRNA. As a result, transcription efficiency (E(transcript)) of LFN, denoted as transgene expression divided by the amount of nuclear pDNA was about 16 times less than that for adenovirus. Furthermore, translation efficiency (E(translate)), denoted as transfection activity divided by mRNA expression was approximately 460 times less in LFN. Imaging of the decondensed form of DNA by in situ hybridization revealed that poor decondensation efficiency of LFN is involved in the inferior E(transcript). Moreover, the inferior translation efficiency (E(translate)) of LFN was mainly due to electrostatic interactions between LFN and mRNA. Collectively, an improvement in nuclear decondensation and the diminution of the interaction between vector and mRNA is essential for the development of new generations of non-viral vectors.
Figures

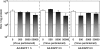

Similar articles
-
Quantitative comparison of intracellular trafficking and nuclear transcription between adenoviral and lipoplex systems.Mol Ther. 2006 Apr;13(4):786-94. doi: 10.1016/j.ymthe.2005.10.007. Epub 2005 Dec 20. Mol Ther. 2006. PMID: 16364692
-
Cell cycle dependent transcription, a determinant factor of heterogeneity in cationic lipid-mediated transgene expression.J Gene Med. 2007 Mar;9(3):197-207. doi: 10.1002/jgm.1010. J Gene Med. 2007. PMID: 17351985
-
Intranuclear fluorescence resonance energy transfer analysis of plasmid DNA decondensation from nonviral gene carriers.J Gene Med. 2009 Jul;11(7):615-23. doi: 10.1002/jgm.1338. J Gene Med. 2009. PMID: 19396931
-
[Development of non-viral vector based on the quantitative comparison of intracellular trafficking with viral vector].Yakugaku Zasshi. 2006 Nov;126(11):1047-57. doi: 10.1248/yakushi.126.1047. Yakugaku Zasshi. 2006. PMID: 17077611 Review. Japanese.
-
[Regulation and analysis of intracellular trafficking of nucleic acids based on quantitative and dynamic imaging].Yakugaku Zasshi. 2011;131(11):1545-56. doi: 10.1248/yakushi.131.1545. Yakugaku Zasshi. 2011. PMID: 22041692 Review. Japanese.
Cited by
-
Low Electric Treatment activates Rho GTPase via Heat Shock Protein 90 and Protein Kinase C for Intracellular Delivery of siRNA.Sci Rep. 2019 Mar 11;9(1):4114. doi: 10.1038/s41598-019-40904-z. Sci Rep. 2019. PMID: 30858501 Free PMC article.
-
Nucleic acid delivery: the missing pieces of the puzzle?Acc Chem Res. 2012 Jul 17;45(7):1153-62. doi: 10.1021/ar3000162. Epub 2012 Mar 19. Acc Chem Res. 2012. PMID: 22428908 Free PMC article. Review.
-
Emerging links between surface nanotechnology and endocytosis: impact on nonviral gene delivery.Nano Today. 2010 Dec 1;5(6):553-569. doi: 10.1016/j.nantod.2010.10.007. Nano Today. 2010. PMID: 21383869 Free PMC article.
-
The novel functional nucleic acid iRed effectively regulates target genes following cytoplasmic delivery by faint electric treatment.Sci Technol Adv Mater. 2016 Sep 16;17(1):554-562. doi: 10.1080/14686996.2016.1221726. eCollection 2016. Sci Technol Adv Mater. 2016. PMID: 27877903 Free PMC article.
-
DNA nuclear targeting sequences for non-viral gene delivery.Pharm Res. 2011 Jul;28(7):1707-22. doi: 10.1007/s11095-011-0407-8. Epub 2011 Mar 18. Pharm Res. 2011. PMID: 21424159 Free PMC article.
References
-
- Dinser R, Kreppel F, Zaucke F, Blank C, Paulsson M, Kochanek S, Maurer P. Comparison of long-term transgene expression after non-viral and adenoviral gene transfer into primary articular chondrocytes. Histochem. Cell. Biol. 2001;116:69–77. - PubMed
-
- Hama S, Akita H, Ito R, Mizuguchi H, Hayakawa T, Harashima H. Quantitative comparison of intracellular trafficking and nuclear transcription between adenoviral and lipoplex systems. Mol. Ther. 2006;13:786–794. - PubMed
-
- Marshall E. Gene therapy death prompts review of adenovirus vector. Science. 1999;286:2244–2245. - PubMed
-
- Hacein-Bey-Abina S, von Kalle C, Schmidt M, Le Deist F, Wulffraat N, McIntyre E, Radford I, Villeval JL, Fraser CC, et al. A serious adverse event after successful gene therapy for X-linked severe combined immunodeficiency. N. Engl. J. Med. 2003;348:255–256. - PubMed
-
- Parker AL, Newman C, Briggs S, Seymour L, Sheridan PJ. Nonviral gene delivery: techniques and implications for molecular medicine. Expert Rev. Mol. Med. 2003;2003:1–15. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

