Roles of volume-sensitive chloride channel in excitotoxic neuronal injury
- PMID: 17287519
- PMCID: PMC6673589
- DOI: 10.1523/JNEUROSCI.4694-06.2007
Roles of volume-sensitive chloride channel in excitotoxic neuronal injury
Abstract
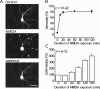
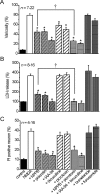
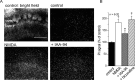
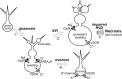
Excitotoxicity is associated with stroke, brain trauma, and a number of neurodegenerative disorders. In the brain, during excitotoxic insults, neurons undergo rapid swelling in both the soma and dendrites. Focal swellings along the dendrites called varicosities are considered to be a hallmark of acute excitotoxic neuronal injury. However, it is not clear what pathway is involved in the neuronal anion flux that leads to the formation and resolution of excitotoxic varicosities. Here, we assessed the roles of the volume-sensitive outwardly rectifying (VSOR) Cl- channel in excitotoxic responses in mouse cortical neurons. Whole-cell patch-clamp recordings revealed that the VSOR Cl- channel in cultured neurons was activated by NMDA exposure. Moreover, robust expression of this channel on varicosities was confirmed by on-cell and nystatin-perforated vesicle patch techniques. VSOR channel blockers, but not blockers of GABA(A) receptors and Cl- transporters, abolished not only varicosity resolution after sublethal excitotoxic stimulation but also necrotic death after sustained varicosity formation induced by prolonged NMDA exposure in cortical neurons. The present slice-patch experiments demonstrated, for the first time, expression of the VSOR Cl- channels in somatosensory pyramidal neurons. NMDA-induced necrotic neuronal death in slice preparations was largely suppressed by a blocker of the VSOR Cl- channel but not of the GABA(A) receptor. These results indicate that VSOR Cl- channels exert dual, reciprocal actions on neuronal excitotoxicity by serving as major anionic pathways both for varicosity recovery after washout of an excitotoxic stimulant and for persistent varicosity formation under prolonged excitotoxic insults leading to necrosis in cortical neurons.
Figures






Similar articles
-
Excitotoxic death induced by released glutamate in depolarized primary cultures of mouse cerebellar granule cells is dependent on GABAA receptors and niflumic acid-sensitive chloride channels.Eur J Neurosci. 2005 Jan;21(1):103-12. doi: 10.1111/j.1460-9568.2004.03848.x. Eur J Neurosci. 2005. PMID: 15654847
-
Distinct roles for sodium, chloride, and calcium in excitotoxic dendritic injury and recovery.Exp Neurol. 1998 Nov;154(1):241-58. doi: 10.1006/exnr.1998.6929. Exp Neurol. 1998. PMID: 9875285
-
Glutamate receptor ion channel properties predict vulnerability to cytotoxicity in a transfected nonneuronal cell line.Mol Cell Neurosci. 1996 Feb;7(2):102-15. doi: 10.1006/mcne.1996.0008. Mol Cell Neurosci. 1996. PMID: 8731479
-
Roles of volume-regulatory anion channels, VSOR and Maxi-Cl, in apoptosis, cisplatin resistance, necrosis, ischemic cell death, stroke and myocardial infarction.Curr Top Membr. 2019;83:205-283. doi: 10.1016/bs.ctm.2019.03.001. Epub 2019 Apr 19. Curr Top Membr. 2019. PMID: 31196606 Review.
-
Similarity of neuronal cell injury and death in AIDS dementia and focal cerebral ischemia: potential treatment with NMDA open-channel blockers and nitric oxide-related species.Brain Pathol. 1996 Oct;6(4):507-17. doi: 10.1111/j.1750-3639.1996.tb00879.x. Brain Pathol. 1996. PMID: 8944320 Review.
Cited by
-
Transient increase in neuronal chloride concentration by neuroactive aminoacids released from glioma cells.Front Mol Neurosci. 2012 Nov 26;5:100. doi: 10.3389/fnmol.2012.00100. eCollection 2012. Front Mol Neurosci. 2012. PMID: 23189038 Free PMC article.
-
The role of PSD-95 and cypin in morphological changes in dendrites following sublethal NMDA exposure.J Neurosci. 2011 Oct 26;31(43):15468-80. doi: 10.1523/JNEUROSCI.2442-11.2011. J Neurosci. 2011. PMID: 22031893 Free PMC article.
-
Chloride channel blocker 4,4-diisothiocyanatostilbene-2,2'-disulfonic acid inhibits nitric oxide-induced apoptosis in cultured rat hippocampal neurons.Neural Regen Res. 2013 Jan 15;8(2):121-6. doi: 10.3969/j.issn.1673-5374.2013.02.003. Neural Regen Res. 2013. PMID: 25206481 Free PMC article.
-
VRACs and other ion channels and transporters in the regulation of cell volume and beyond.Nat Rev Mol Cell Biol. 2016 May;17(5):293-307. doi: 10.1038/nrm.2016.29. Epub 2016 Apr 1. Nat Rev Mol Cell Biol. 2016. PMID: 27033257 Review.
-
Time dependence in diffusion MRI predicts tissue outcome in ischemic stroke patients.Magn Reson Med. 2021 Aug;86(2):754-764. doi: 10.1002/mrm.28743. Epub 2021 Mar 23. Magn Reson Med. 2021. PMID: 33755261 Free PMC article.
References
-
- Babot Z, Cristófol R, Suñol C. Excitotoxic death induced by released glutamate in depolarized primary cultures of mouse cerebellar granule cells is dependent on GABAA receptors and niflumic acid-sensitive chloride channels. Eur J Neurosci. 2005;21:103–112. - PubMed
-
- Barros LF, Hermosilla T, Castro J. Necrotic volume increase and the early physiology of necrosis. Comp Biochem Physiol A Mol Integr Physiol. 2001;130:401–409. - PubMed
-
- Chebabo SR, Hester MA, Aitken PG, Somjen GG. Hypotonic exposure enhances synaptic transmission and triggers spreading depression in rat hippocampal tissue slices. Brain Res. 1995;695:203–216. - PubMed
-
- Chen Q, Moulder K, Tenkova T, Hardy K, Olney JW, Romano C. Excitotoxic cell death dependent on inhibitory receptor activation. Exp Neurol. 1999;160:215–225. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
