cAMP microdomains and L-type Ca2+ channel regulation in guinea-pig ventricular myocytes
- PMID: 17289786
- PMCID: PMC2075464
- DOI: 10.1113/jphysiol.2006.124891
cAMP microdomains and L-type Ca2+ channel regulation in guinea-pig ventricular myocytes
Abstract
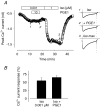
Many different receptors can stimulate cAMP synthesis in the heart, but not all elicit the same functional responses. For example, it has been recognized for some time that prostaglandins such as PGE1 increase cAMP production and activate PKA, but they do not elicit responses like those produced by beta-adrenergic receptor (betaAR) agonists such as isoproterenol (isoprenaline), even though both stimulate the same signalling pathway. In the present study, we confirm that isoproterenol, but not PGE1, is able to produce cAMP-dependent stimulation of the L-type Ca(2+) current in guinea pig ventricular myocytes. This is despite finding evidence that these cells express EP(4) prostaglandin receptors, which are known to activate G(s)-dependent signalling pathways. Using fluorescence resonance energy transfer-based biosensors that are either freely diffusible or bound to A kinase anchoring proteins, we demonstrate that the difference is due to the ability of isoproterenol to stimulate cAMP production in cytosolic and caveolar compartments of intact cardiac myocytes, while PGE1 only stimulates cAMP production in the cytosolic compartment. Unlike other receptor-mediated responses, compartmentation of PGE1 responses was not due to concurrent activation of a G(i)-dependent signalling pathway or phosphodiesterase activity. Instead, compartmentation of the PGE1 response in cardiac myocytes appears to be due to transient stimulation of cAMP in a microdomain that can communicate directly with the bulk cytosolic compartment but not the caveolar compartment associated with betaAR regulation of L-type Ca(2+) channel function.
Figures



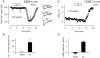

References
-
- Alloatti G, Serazzi L, Levi RC. Prostaglandin I2 (PGI2) enhances calcium current in guinea-pig ventricular heart cells. J Mol Cell Cardiol. 1991;23:851–860. - PubMed
-
- Bers DM. Excitation-Contraction Coupling and Cardiac Contractile Force. 2. Kluwer: Dordrecht; 2001.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

