D2-like dopamine receptors mediate the response to amphetamine in a mouse model of ADHD
- PMID: 17291774
- PMCID: PMC1876677
- DOI: 10.1016/j.nbd.2006.12.011
D2-like dopamine receptors mediate the response to amphetamine in a mouse model of ADHD
Abstract
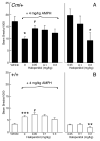
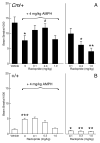
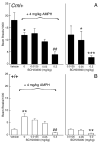
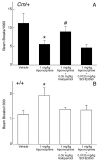
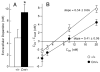
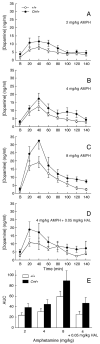
The mechanisms underlying the effects of psychostimulants in attention deficit hyperactivity disorder (ADHD) are not well understood, but indirect evidence implicates D2 dopamine receptors. Here we dissect the components of dopaminergic neurotransmission in the hyperactive mouse mutant coloboma to identify pre- and postsynaptic elements essential for the effects of amphetamine in these mice. Amphetamine treatment reduced locomotor activity in coloboma mice, but induced a robust increase in dopamine overflow suggesting that abnormal regulation of dopamine efflux does not account for the behavioral effect. However, the D2-like dopamine receptor antagonists haloperidol and raclopride, but not the D1-like dopamine receptor antagonist SCH23390, blocked the amphetamine-induced reduction in locomotor activity in coloboma mice, providing direct evidence that D2-like dopamine receptors mediate the effect of amphetamine in these mice. With the precedent established that it is possible to directly antagonize this response, this strategy should prove useful for identifying novel therapeutics in ADHD.
Figures

Similar articles
-
D2 dopamine receptor subtype-mediated hyperactivity and amphetamine responses in a model of ADHD.Neurobiol Dis. 2010 Jan;37(1):228-36. doi: 10.1016/j.nbd.2009.10.009. Epub 2009 Oct 22. Neurobiol Dis. 2010. PMID: 19840852 Free PMC article.
-
CB1 receptor knockout mice display reduced ethanol-induced conditioned place preference and increased striatal dopamine D2 receptors.Neuropsychopharmacology. 2005 Feb;30(2):339-49. doi: 10.1038/sj.npp.1300568. Neuropsychopharmacology. 2005. PMID: 15383833
-
Enhancement of postsynaptic sensitivity to dopaminergic agonists induced by neonatal hippocampal lesions.Neuropsychopharmacology. 1997 Apr;16(4):259-68. doi: 10.1016/S0893-133X(96)00217-5. Neuropsychopharmacology. 1997. PMID: 9094143
-
Dopamine ligands and the stimulus effects of amphetamine: animal models versus human laboratory data.Psychopharmacology (Berl). 1997 Mar;130(1):2-13. doi: 10.1007/s002130050207. Psychopharmacology (Berl). 1997. PMID: 9089844 Review.
-
Coloboma mouse mutant as an animal model of hyperkinesis and attention deficit hyperactivity disorder.Neurosci Biobehav Rev. 2000 Jan;24(1):51-7. doi: 10.1016/s0149-7634(99)00064-0. Neurosci Biobehav Rev. 2000. PMID: 10654661 Review.
Cited by
-
The efficacy of atomoxetine for the treatment of children and adolescents with attention-deficit/hyperactivity disorder: a comprehensive review of over a decade of clinical research.CNS Drugs. 2015 Feb;29(2):131-51. doi: 10.1007/s40263-014-0224-9. CNS Drugs. 2015. PMID: 25698145 Review.
-
Stress, caffeine and ethanol trigger transient neurological dysfunction through shared mechanisms in a mouse calcium channelopathy.Neurobiol Dis. 2013 Feb;50:151-9. doi: 10.1016/j.nbd.2012.09.005. Epub 2012 Sep 23. Neurobiol Dis. 2013. PMID: 23009754 Free PMC article.
-
D2 dopamine receptor subtype-mediated hyperactivity and amphetamine responses in a model of ADHD.Neurobiol Dis. 2010 Jan;37(1):228-36. doi: 10.1016/j.nbd.2009.10.009. Epub 2009 Oct 22. Neurobiol Dis. 2010. PMID: 19840852 Free PMC article.
-
Cortico-basal ganglia circuits underlying dysfunctional control of motor behaviors in neuropsychiatric disorders.Curr Opin Genet Dev. 2020 Dec;65:151-159. doi: 10.1016/j.gde.2020.05.042. Epub 2020 Jul 17. Curr Opin Genet Dev. 2020. PMID: 32688249 Free PMC article. Review.
-
Functional analysis of dopaminergic systems in a DYT1 knock-in mouse model of dystonia.Neurobiol Dis. 2012 Oct;48(1):66-78. doi: 10.1016/j.nbd.2012.05.009. Epub 2012 May 31. Neurobiol Dis. 2012. PMID: 22659308 Free PMC article.
References
-
- Archer T, Fredriksson A, Jonsson G, Lewander T, Mohammed AK, Ross SB, Soderberg U. Central noradrenaline depletion antagonizes aspects of d-amphetamine-induced hyperactivity in the rat. Psychopharmacology. 1986;88:141–146. - PubMed
-
- Arnold LE, Christopher J, Huestis R, Smeltzer DJ. Methylphenidate vs dextroamphetamine vs caffeine in minimal brain dysfunction. Arch Gen Psychiatry. 1978;35:463–473. - PubMed
-
- Bamford NS, Zhang H, Schmitz Y, Wu NP, Cepeda C, Levine MS, Schmauss C, Zakharenko SS, Zablow L, Sulzer D. Heterosynaptic dopamine neurotransmission selects sets of corticostriatal terminals. Neuron. 2004;42:653–663. - PubMed
-
- Barr CL, Feng Y, Wigg K, Bloom S, Roberts W, Malone M, Schachar R, Tannock R, Kennedy JL. Identification of DNA variants in the SNAP-25 gene and linkage study of these polymorphisms and attention-deficit hyperactivity disorder. Mol Psychiatry. 2000;5:405–409. - PubMed
-
- Bradley C. The behavior of children receiving Benzedrine. Am J Psychiatry. 1937;94:577–585.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

