'Nicotine deprivation effect' in rats with intermittent 23-hour access to intravenous nicotine self-administration
- PMID: 17292952
- PMCID: PMC2386418
- DOI: 10.1016/j.pbb.2007.01.004
'Nicotine deprivation effect' in rats with intermittent 23-hour access to intravenous nicotine self-administration
Abstract
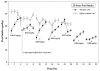
Our previous work demonstrates that rats allowed extended 23-hour access to intravenous nicotine self-administration (IVSA) display voluntary, dose-related levels of nicotine intake (i.e., higher doses result in higher intake) that remain stable across 40 days. This study examined whether an escalating dose regimen with intermittent abstinence periods produces different levels of nicotine intake relative to those observed during continuous access to a fixed unit dose. Rats were trained to nose-poke for food and water in 23-hour sessions prior to and after recovery from surgical implantation of jugular catheters. Animals (n=12) then were given access to nicotine IVSA in 4-day cycles, each separated by three intervening days of abstinence in their home cage. The unit dose available for nicotine IVSA was increased between cycles as follows: 0.015, 0.03, 0.06, 0.09 mg/kg/0.1 ml infusion/1 s, fixed ratio 1. Control rats (n=6) were given access to saline for five 4-day IVSA periods. Nicotine dependence was assessed by examining physical signs of withdrawal following an injection of the nicotinic antagonist mecamylamine (1.5 mg/kg, i.p.). Nicotine intake dose-dependently increased between cycles. Within each cycle, nicotine intake was highest on the first day after abstinence and decreased over the next 3 days of continuous access. Mecamylamine produced a significant increase in overt signs of withdrawal in the 23-hour access animals comparable to that observed in previous studies of nicotine dependence. Our findings suggest that abstinence from nicotine may produce a "deprivation effect" in nicotine-dependent rats. In addition, intermittent access to increasing unit doses appears to produce higher levels of nicotine intake than continuous access to a constant unit nicotine dose.
Figures


References
-
- Balfour DJ. Neural mechanisms underlying nicotine dependence. Addiction. 1994;89:1419–1423. - PubMed
-
- Becker HC, Baros AM. Effect of duration and pattern of chronic ethanol exposure on tolerance to the discriminative stimulus effects of ethanol in C57BL/6J mice. J Pharmacol Exp Ther. 2006 in press. - PubMed
-
- Benowitz NL, Jacob P., 3rd Nicotine and carbon monoxide intake from high- and low-yield cigarettes. Clin Pharmacol Ther. 1984;36:265–270. - PubMed
-
- Benwell ME, Balfour DJ, Anderson JM. Evidence that tobacco smoking increases the density of (−)-[3H]nicotine binding sites in human brain. J Neurochem. 1988;50:1243–1247. - PubMed
-
- Caine SB, Lintz R, Koob GF. Intravenous drug self-administration techniques in animals. In: Sahgal A, editor. Behavioural Neuroscience: A Practical Approach. Vol. 2. IRL Press; Oxford: 1993. pp. 117–143.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

