Nonhematopoietic NADPH oxidase regulation of lung eosinophilia and airway hyperresponsiveness in experimentally induced asthma
- PMID: 17293377
- PMCID: PMC2710034
- DOI: 10.1152/ajplung.00208.2006
Nonhematopoietic NADPH oxidase regulation of lung eosinophilia and airway hyperresponsiveness in experimentally induced asthma
Abstract
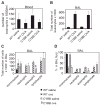
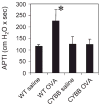
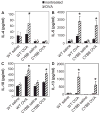
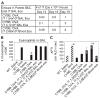
Pulmonary eosinophilia is one of the most consistent hallmarks of asthma. Infiltration of eosinophils into the lung in experimental asthma is dependent on the adhesion molecule vascular cell adhesion molecule-1 (VCAM-1) on endothelial cells. Ligation of VCAM-1 activates endothelial cell NADPH oxidase, which is required for VCAM-1-dependent leukocyte migration in vitro. To examine whether endothelial-derived NADPH oxidase modulates eosinophil recruitment in vivo, mice deficient in NADPH oxidase (CYBB mice) were irradiated and received wild-type hematopoietic cells to generate chimeric CYBB mice. In response to ovalbumin (OVA) challenge, the chimeric CYBB mice had increased numbers of eosinophils bound to the endothelium as well as reduced eosinophilia in the lung tissue and bronchoalveolar lavage. This occurred independent of changes in VCAM-1 expression, cytokine/chemokine levels (IL-5, IL-10, IL-13, IFNgamma, or eotaxin), or numbers of T cells, neutrophils, or mononuclear cells in the lavage fluids or lung tissue of OVA-challenged mice. Importantly, the OVA-challenged chimeric CYBB mice had reduced airway hyperresponsiveness (AHR). The AHR in OVA-challenged chimeric CYBB mice was restored by bypassing the endothelium with intratracheal administration of eosinophils. These data suggest that VCAM-1 induction of NADPH oxidase in the endothelium is necessary for the eosinophil recruitment during allergic inflammation. Moreover, these studies provide a basis for targeting VCAM-1-dependent signaling pathways in asthma therapies.
Figures





References
-
- Al-Mehdi AB, Zhao G, Dodia C, Tozawa K, Costa K, Muzykantov V, Ross C, Blecha F, Dinauer M, Fisher AB. Endothelial NADPH oxidase as the source of oxidants in lungs exposed to ischemia or high K+ Circ Res. 1998;83:730–737. - PubMed
-
- Bankers-Fulbright JL, Kita H, Gleich GJ, O’Grady SM. Regulation of human eosinophil NADPH oxidase activity: a central role for PKC delta. J Cell Physiol. 2001;189:306–315. - PubMed
-
- Bjorgvinsdottir H, Ding C, Pech N, Gifford MA, Li LL, Dinauer MC. Retroviral-mediated gene transfer of gp91phox into bone marrow cells rescues defect in host defense against Aspergillus fumigatus in murine X-linked chronic granulomatous disease. Blood. 1997;89:41–48. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

