Dominant-negative modification reveals the regulatory function of the multimeric cysteine synthase protein complex in transgenic tobacco
- PMID: 17293569
- PMCID: PMC1867341
- DOI: 10.1105/tpc.106.043125
Dominant-negative modification reveals the regulatory function of the multimeric cysteine synthase protein complex in transgenic tobacco
Abstract
Cys synthesis in plants constitutes the entry of reduced sulfur from assimilatory sulfate reduction into metabolism. The catalyzing enzymes serine acetyltransferase (SAT) and O-acetylserine (OAS) thiol lyase (OAS-TL) reversibly form the heterooligomeric Cys synthase complex (CSC). Dominant-negative mutation of the CSC showed the crucial function for the regulation of Cys biosynthesis in vivo. An Arabidopsis thaliana SAT was overexpressed in the cytosol of transgenic tobacco (Nicotiana tabacum) plants in either enzymatically active or inactive forms that were both shown to interact efficiently with endogenous tobacco OAS-TL proteins. Active SAT expression resulted in a 40-fold increase in SAT activity and strong increases in the reaction intermediate OAS as well as Cys, glutathione, Met, and total sulfur contents. However, inactive SAT expression produced much greater enhancing effects, including 30-fold increased Cys levels, attributable, apparently, to the competition of inactive transgenic SAT with endogenous tobacco SAT for binding to OAS-TL. Expression levels of tobacco SAT and OAS-TL remained unaffected. Flux control coefficients suggested that the accumulation of OAS and Cys in both types of transgenic plants was accomplished by different mechanisms. These data provide evidence that the CSC and its subcellular compartmentation play a crucial role in the control of Cys biosynthesis, a unique function for a plant metabolic protein complex.
Figures
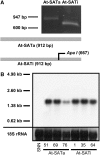


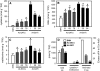



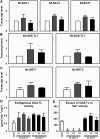
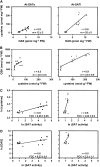

References
-
- Berkowitz, O., Wirtz, M., Wolf, A., Kuhlmann, J., and Hell, R. (2002). Use of biomolecular interaction analysis to elucidate the regulatory mechanism of the cysteine synthase complex from Arabidopsis thaliana. J. Biol. Chem. 277 30629–30634. - PubMed
-
- Blaszczyk, A., Brodzik, R., and Sirko, A. (1999). Increased resistance to oxidative stress in transgenic tobacco plants overexpressing bacterial serine acetyltransferase. Plant J. 20 237–243. - PubMed
-
- Bogdanova, N., Bork, C., and Hell, R. (1995). Cysteine biosynthesis in plants: Isolation and functional identification of a cDNA encoding a serine acetyltransferase from Arabidopsis thaliana. FEBS Lett. 358 43–47. - PubMed
-
- Bogdanova, N., and Hell, R. (1997). Cysteine synthesis in plants: Protein-protein interactions of serine acetyltransferase from Arabidopsis thaliana. Plant J. 11 251–262. - PubMed
-
- Boorer, K.J., Frommer, W.B., Bush, D.R., Kreman, M., Loo, D.D., and Wright, E.M. (1996). Kinetics and specificity of a H+/amino acid transporter from Arabidopsis thaliana. J. Biol. Chem. 271 2213–2220. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

