Vesicular release of glutamate from unmyelinated axons in white matter
- PMID: 17293857
- PMCID: PMC2140234
- DOI: 10.1038/nn1854
Vesicular release of glutamate from unmyelinated axons in white matter
Abstract
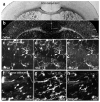
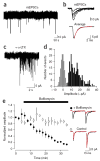
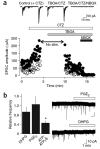
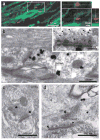
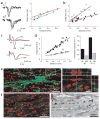
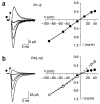
Directed fusion of transmitter-laden vesicles enables rapid intercellular signaling in the central nervous system and occurs at synapses within gray matter. Here we show that action potentials also induce the release of glutamate from axons in the corpus callosum, a white matter region responsible for interhemispheric communication. Callosal axons release glutamate by vesicular fusion, which induces quantal AMPA receptor-mediated currents in NG2(+) glial progenitors at anatomically distinct axo-glial synaptic junctions. Glutamate release from axons was facilitated by repetitive stimulation and could be inhibited through activation of metabotropic autoreceptors. Although NG2(+) cells form associations with nodes of Ranvier in white matter, measurements of conduction velocity indicated that unmyelinated fibers are responsible for glutamatergic signaling with NG2(+) glia. This activity-dependent secretion of glutamate was prevalent in the developing and mature mouse corpus callosum, indicating that axons within white matter both conduct action potentials and engage in rapid neuron-glia communication.
Conflict of interest statement
COMPETING INTERESTS STATEMENT
The authors declare that they have no competing financial interests.
Figures
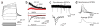

Comment in
-
Surprising synapses deep in the brain.Nat Neurosci. 2007 Mar;10(3):267-9. doi: 10.1038/nn0307-267. Nat Neurosci. 2007. PMID: 17318215 No abstract available.
Similar articles
-
Axons and astrocytes release ATP and glutamate to evoke calcium signals in NG2-glia.Glia. 2010 Jan 1;58(1):66-79. doi: 10.1002/glia.20902. Glia. 2010. PMID: 19533604
-
Vesicular glutamate release from axons in white matter.Nat Neurosci. 2007 Mar;10(3):311-20. doi: 10.1038/nn1850. Epub 2007 Feb 11. Nat Neurosci. 2007. PMID: 17293860
-
The fate of synaptic input to NG2 glial cells: neurons specifically downregulate transmitter release onto differentiating oligodendroglial cells.J Neurosci. 2010 Jun 16;30(24):8320-31. doi: 10.1523/JNEUROSCI.0854-10.2010. J Neurosci. 2010. PMID: 20554883 Free PMC article.
-
Synaptic signaling between neurons and glia.Glia. 2004 Aug 15;47(3):290-298. doi: 10.1002/glia.20060. Glia. 2004. PMID: 15252819 Review.
-
Neuron-glia synapses in the brain.Brain Res Rev. 2010 May;63(1-2):130-7. doi: 10.1016/j.brainresrev.2009.12.003. Epub 2009 Dec 16. Brain Res Rev. 2010. PMID: 20018210 Free PMC article. Review.
Cited by
-
Myelin status and oligodendrocyte lineage cells over time after spinal cord injury: What do we know and what still needs to be unwrapped?Glia. 2019 Nov;67(11):2178-2202. doi: 10.1002/glia.23702. Epub 2019 Aug 24. Glia. 2019. PMID: 31444938 Free PMC article. Review.
-
L-Type Ca2+ Channels of NG2 Glia Determine Proliferation and NMDA Receptor-Dependent Plasticity.Front Cell Dev Biol. 2021 Oct 21;9:759477. doi: 10.3389/fcell.2021.759477. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34746151 Free PMC article.
-
NMDA Receptors in Glial Cells: Pending Questions.Curr Neuropharmacol. 2013 May;11(3):250-62. doi: 10.2174/1570159X11311030002. Curr Neuropharmacol. 2013. PMID: 24179462 Free PMC article.
-
Neuron-NG2 cell synapses: novel functions for regulating NG2 cell proliferation and differentiation.Biomed Res Int. 2013;2013:402843. doi: 10.1155/2013/402843. Epub 2013 Aug 1. Biomed Res Int. 2013. PMID: 23984358 Free PMC article. Review.
-
Electrophysiological properties of NG2(+) cells: Matching physiological studies with gene expression profiles.Brain Res. 2016 May 1;1638(Pt B):138-160. doi: 10.1016/j.brainres.2015.09.010. Epub 2015 Sep 15. Brain Res. 2016. PMID: 26385417 Free PMC article. Review.
References
-
- Clancy B, Silva-Filho M, Friedlander MJ. Structure and projections of white matter neurons in the postnatal rat visual cortex. J Comp Neurol. 2001;434:233–252. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

