Chk1-mediated phosphorylation of FANCE is required for the Fanconi anemia/BRCA pathway
- PMID: 17296736
- PMCID: PMC1899922
- DOI: 10.1128/MCB.02357-06
Chk1-mediated phosphorylation of FANCE is required for the Fanconi anemia/BRCA pathway
Abstract
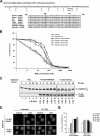
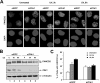
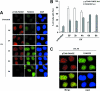
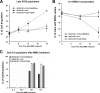
The eleven Fanconi anemia (FA) proteins cooperate in a novel pathway required for the repair of DNA cross-links. Eight of the FA proteins (A, B, C, E, F, G, L, and M) form a core enzyme complex, required for the monoubiquitination of FANCD2 and the assembly of FANCD2 nuclear foci. Here, we show that, in response to DNA damage, Chk1 directly phosphorylates the FANCE subunit of the FA core complex on two conserved sites (threonine 346 and serine 374). Phosphorylated FANCE assembles in nuclear foci and colocalizes with FANCD2. A nonphosphorylated mutant form of FANCE (FANCE-T346A/S374A), when expressed in a FANCE-deficient cell line, allows FANCD2 monoubiquitination, FANCD2 foci assembly, and normal S-phase progression. However, the mutant FANCE protein fails to complement the mitomycin C hypersensitivity of the transfected cells. Taken together, these results elucidate a novel role of Chk1 in the regulation of the FA/BRCA pathway and in DNA cross-link repair. Chk1-mediated phosphorylation of FANCE is required for a function independent of FANCD2 monoubiquitination.
Figures


References
-
- Auerbach, A. D. 1993. Fanconi anemia diagnosis and the diepoxybutane (DEB) test. Exp. Hematol. 21:731-733. - PubMed
-
- de Winter, J. P., F. Leveille, C. G. van Berkel, M. A. Rooimans, L. van Der Weel, J. Steltenpool, I. Demuth, N. V. Morgan, N. Alon, L. Bosnoyan-Collins, J. Lightfoot, P. A. Leegwater, Q. Waisfisz, K. Komatsu, F. Arwert, J. C. Pronk, C. G. Mathew, M. Digweed, M. Buchwald, and H. Joenje. 2000. Isolation of a cDNA representing the Fanconi anemia complementation group E gene. Am. J. Hum. Genet. 67:1306-1308. - PMC - PubMed
-
- Dunn, J., M. Potter, A. Rees, and T. M. Runger. 2006. Activation of the Fanconi anemia/BRCA pathway and recombination repair in the cellular response to solar ultraviolet light. Cancer Res. 66:11140-11147. - PubMed
-
- Garcia-Higuera, I., T. Taniguchi, S. Ganesan, M. S. Meyn, C. Timmers, J. Hejna, M. Grompe, and A. D. D'Andrea. 2001. Interaction of the Fanconi anemia proteins and BRCA1 in a common pathway. Mol. Cell 7:249-262. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
