Treponema pallidum elicits innate and adaptive cellular immune responses in skin and blood during secondary syphilis: a flow-cytometric analysis
- PMID: 17299719
- PMCID: PMC2131710
- DOI: 10.1086/511822
Treponema pallidum elicits innate and adaptive cellular immune responses in skin and blood during secondary syphilis: a flow-cytometric analysis
Abstract
Background: Syphilis is caused by the spirochetal pathogen Treponema pallidum. The local and systemic cellular immune responses elicited by the bacterium have not been well studied in humans.
Methods: We used multiparameter flow cytometry to characterize leukocyte immunophenotypes in skin and peripheral blood from 23 patients with secondary syphilis and 5 healthy control subjects recruited in Cali, Colombia. Dermal leukocytes were obtained from fluid aspirated from epidermal suction blisters raised over secondary syphilis skin lesions.
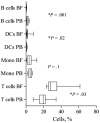
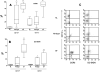
Results: Compared with peripheral blood (PB), blister fluids (BFs) were enriched for CD4(+) and CD8(+) T cells, activated monocytes/macrophages, and CD11c(+) monocytoid and CD11c(-) plasmacytoid dendritic cells (mDCs and pDCs, respectively). Nearly all mDCs in BFs expressed the human immunodeficiency virus (HIV) coreceptors CCR5 and DC-specific intercellular adhesion molecule 3-grabbing nonintegrin (DC-SIGN) and high levels of human leukocyte antigen (HLA)-DR. Dermal pDCs expressed both HIV coreceptors without increases in HLA-DR intensity. Compared with normal blood, circulating mDCs in patients with syphilis expressed higher levels of both CCR5 and DC-SIGN, whereas circulating pDCs in patients expressed only higher levels of DC-SIGN. Most dermal T cells were CCR5(+) and displayed a memory (CD27(+)/CD45RO(+)) or memory/effector (CD27(-)/CD45RO(+)) immunophenotype. A corresponding shift toward memory and memory/effector immunophenotype was clearly discernible among circulating CD4(+) T cells. Compared with PB from control subjects, a larger percentage of CD4(+) T cells in PB from patients with syphilis expressed the activation markers CD69 and CD38.
Conclusions: During secondary syphilis, T. pallidum simultaneously elicits local and systemic innate and adaptive immune responses that may set the stage for the bidirectional transmission of HIV.
Conflict of interest statement
Potential conflicts of interest: none reported.
Figures
Similar articles
-
The cutaneous response in humans to Treponema pallidum lipoprotein analogues involves cellular elements of both innate and adaptive immunity.J Immunol. 2001 Mar 15;166(6):4131-40. doi: 10.4049/jimmunol.166.6.4131. J Immunol. 2001. PMID: 11238663
-
Coevolution of markers of innate and adaptive immunity in skin and peripheral blood of patients with erythema migrans.J Immunol. 2003 Sep 1;171(5):2660-70. doi: 10.4049/jimmunol.171.5.2660. J Immunol. 2003. PMID: 12928420
-
Immune evasion and recognition of the syphilis spirochete in blood and skin of secondary syphilis patients: two immunologically distinct compartments.PLoS Negl Trop Dis. 2012;6(7):e1717. doi: 10.1371/journal.pntd.0001717. Epub 2012 Jul 17. PLoS Negl Trop Dis. 2012. PMID: 22816000 Free PMC article.
-
The immune response to infection with Treponema pallidum, the stealth pathogen.Microbes Infect. 2002 Sep;4(11):1133-40. doi: 10.1016/s1286-4579(02)01638-6. Microbes Infect. 2002. PMID: 12361913 Review.
-
A double-edged sword: does highly active antiretroviral therapy contribute to syphilis incidence by impairing immunity to Treponema pallidum?Sex Transm Infect. 2017 Aug;93(5):374-378. doi: 10.1136/sextrans-2016-052870. Epub 2017 Jan 16. Sex Transm Infect. 2017. PMID: 28093460 Free PMC article. Review.
Cited by
-
Immunity to Sexually Transmitted Bacterial Infections of the Female Genital Tract: Toward Effective Vaccines.Vaccines (Basel). 2024 Aug 1;12(8):863. doi: 10.3390/vaccines12080863. Vaccines (Basel). 2024. PMID: 39203989 Free PMC article. Review.
-
Investigation of the immune escape mechanism of Treponema pallidum.Infection. 2023 Apr;51(2):305-321. doi: 10.1007/s15010-022-01939-z. Epub 2022 Oct 19. Infection. 2023. PMID: 36260281 Review.
-
Pathogenic Leptospires Limit Dendritic Cell Activation Through Avoidance of TLR4 and TRIF Signaling.Front Immunol. 2022 Jun 22;13:911778. doi: 10.3389/fimmu.2022.911778. eCollection 2022. Front Immunol. 2022. PMID: 35812397 Free PMC article.
-
Potential use of saliva infrared spectra and machine learning for a minimally invasive screening test for congenital syphilis in infants.Sci Rep. 2025 Jul 1;15(1):22412. doi: 10.1038/s41598-025-05144-4. Sci Rep. 2025. PMID: 40594373 Free PMC article.
-
Haemophilus ducreyi partially activates human myeloid dendritic cells.Infect Immun. 2007 Dec;75(12):5678-85. doi: 10.1128/IAI.00702-07. Epub 2007 Oct 8. Infect Immun. 2007. PMID: 17923525 Free PMC article.
References
-
- Radolf JD, Lukehart SA. Immunology of syphilis. In: Radolf JD, Lukehart SA, editors. Pathogenic treponemes: cellular and molecular biology. Norfolk, UK: Caister Academic Press; 2006. pp. 285–322.
-
- Rompalo AM, Lawlor J, Seaman P, Quinn TC, Zenilman JM, Hook EW., III Modification of syphilitic genital ulcer manifestations by coexistent HIV infection. Sex Transm Dis. 2001;28:448–54. - PubMed
-
- Quinn TC, Glasser D, Cannon RO, et al. Human immunodeficiency virus infection among patients attending clinics for sexually transmitted diseases. N Engl J Med. 1988;318:197–203. - PubMed
-
- Sellati TJ, Waldrop SL, Salazar JC, Bergstresser PR, Picker LJ, Radolf JD. The cutaneous response in humans to Treponema pallidum lipoprotein analogues involves cellular elements of both innate and adaptive immunity. J Immunol. 2001;166:4131–40. - PubMed
-
- Theus SA, Harrich DA, Gaynor R, Radolf JD, Norgard MV. Treponema pallidum, treponemal lipoproteins, and synthetic lipoprotein analogs (lipopeptides) induce human immunodeficiency virus-1 gene expression in monocyte/macrophages via nuclear translocation of NF-κB. J Infect Dis. 1998;177:941–50. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

