The SARS-Coronavirus Membrane protein induces apoptosis through modulating the Akt survival pathway
- PMID: 17306213
- PMCID: PMC7094499
- DOI: 10.1016/j.abb.2007.01.012
The SARS-Coronavirus Membrane protein induces apoptosis through modulating the Akt survival pathway
Abstract
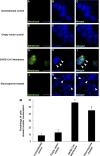
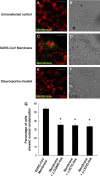
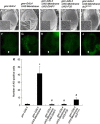
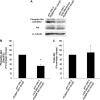
A number of viral gene products are capable of triggering apoptotic cell death through interfering with cellular signaling cascades, including the Akt kinase pathway. In this study, the pro-apoptotic role of the SARS-CoV Membrane (M) structural protein is described. We found that the SARS-CoV M protein induced apoptosis in both HEK293T cells and transgenic Drosophila. We further showed that M protein-induced apoptosis involved mitochondrial release of cytochrome c protein, and could be suppressed by caspase inhibitors. Over-expression of M caused a dominant rough-eye phenotype in adult Drosophila. By performing a forward genetic modifier screen, we identified phosphoinositide-dependent kinase-1 (PDK-1) as a dominant suppressor of M-induced apoptotic cell death. Both PDK-1 and Akt kinases play essential roles in the cell survival signaling pathway. Altogether, our data show that SARS-CoV M protein induces apoptosis through the modulation of the cellular Akt pro-survival pathway and mitochondrial cytochrome c release.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

