Replication history of B lymphocytes reveals homeostatic proliferation and extensive antigen-induced B cell expansion
- PMID: 17312005
- PMCID: PMC2137914
- DOI: 10.1084/jem.20060964
Replication history of B lymphocytes reveals homeostatic proliferation and extensive antigen-induced B cell expansion
Abstract
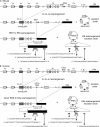
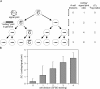
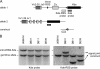
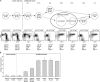
The contribution of proliferation to B lymphocyte homeostasis and antigen responses is largely unknown. We quantified the replication history of mouse and human B lymphocyte subsets by calculating the ratio between genomic coding joints and signal joints on kappa-deleting recombination excision circles (KREC) of the IGK-deleting rearrangement. This approach was validated with in vitro proliferation studies. We demonstrate that naive mature B lymphocytes, but not transitional B lymphocytes, undergo in vivo homeostatic proliferation in the absence of somatic mutations in the periphery. T cell-dependent B cell proliferation was substantially higher and showed higher frequencies of somatic hypermutation than T cell-independent responses, fitting with the robustness and high affinity of T cell-dependent antibody responses. More extensive proliferation and somatic hypermutation in antigen-experienced B lymphocytes from human adults compared to children indicated consecutive responses upon additional antigen exposures. Our combined observations unravel the contribution of proliferation to both B lymphocyte homeostasis and antigen-induced B cell expansion. We propose an important role for both processes in humoral immunity. These new insights will support the understanding of peripheral B cell regeneration after hematopoietic stem cell transplantation or B cell-directed antibody therapy, and the identification of defects in homeostatic or antigen-induced B cell proliferation in patients with common variable immunodeficiency or another antibody deficiency.
Figures


Similar articles
-
Real-time quantitative (RQ-)PCR approach to quantify the contribution of proliferation to B lymphocyte homeostasis.Methods Mol Biol. 2013;979:133-45. doi: 10.1007/978-1-62703-290-2_11. Methods Mol Biol. 2013. PMID: 23397393
-
Naive B lymphocytes undergo homeostatic proliferation in response to B cell deficit.J Immunol. 2002 Dec 15;169(12):6795-805. doi: 10.4049/jimmunol.169.12.6795. J Immunol. 2002. PMID: 12471111
-
Acquisition of innate B cell properties and generation of autoreactive IgA antibodies by follicular B cells during homeostatic proliferation.Front Immunol. 2025 Jan 22;16:1506628. doi: 10.3389/fimmu.2025.1506628. eCollection 2025. Front Immunol. 2025. PMID: 39911392 Free PMC article.
-
Homeostatic and maturation-associated proliferation in the peripheral B-cell compartment.Cell Cycle. 2007 Dec 1;6(23):2890-5. doi: 10.4161/cc.6.23.4952. Cell Cycle. 2007. PMID: 18156800 Review.
-
Non-antigen signals for B-cell growth and differentiation to antibody secretion.Curr Opin Immunol. 1993 Jun;5(3):439-44. doi: 10.1016/0952-7915(93)90066-2. Curr Opin Immunol. 1993. PMID: 7688518 Review.
Cited by
-
Effects of combined antiretroviral therapy on B- and T-cell release from production sites in long-term treated HIV-1+ patients.J Transl Med. 2012 May 16;10:94. doi: 10.1186/1479-5876-10-94. J Transl Med. 2012. PMID: 22591651 Free PMC article.
-
Congenital B cell lymphocytosis explained by novel germline CARD11 mutations.J Exp Med. 2012 Nov 19;209(12):2247-61. doi: 10.1084/jem.20120831. Epub 2012 Nov 5. J Exp Med. 2012. PMID: 23129749 Free PMC article.
-
Utilization of TREC and KREC quantification for the monitoring of early T- and B-cell neogenesis in adult patients after allogeneic hematopoietic stem cell transplantation.J Transl Med. 2013 Aug 14;11:188. doi: 10.1186/1479-5876-11-188. J Transl Med. 2013. PMID: 23941115 Free PMC article.
-
Plasma cells are not restricted to the CD27+ phenotype: characterization of CD27-CD43+ antibody-secreting cells.Front Immunol. 2023 Jul 10;14:1165936. doi: 10.3389/fimmu.2023.1165936. eCollection 2023. Front Immunol. 2023. PMID: 37492569 Free PMC article.
-
Loss of B cells and their precursors is the most constant feature of GATA-2 deficiency in childhood myelodysplastic syndrome.Haematologica. 2016 Jun;101(6):707-16. doi: 10.3324/haematol.2015.137711. Epub 2016 Mar 24. Haematologica. 2016. PMID: 27013649 Free PMC article.
References
-
- Ghia, P., E. ten Boekel, A.G. Rolink, and F. Melchers. 1998. B-cell development: a comparison between mouse and man. Immunol. Today. 19:480–485. - PubMed
-
- Tonegawa, S. 1983. Somatic generation of antibody diversity. Nature. 302:575–581. - PubMed
-
- Thomas, M.D., B. Srivastava, and D. Allman. 2006. Regulation of peripheral B cell maturation. Cell. Immunol. 239:92–102. - PubMed
-
- Allman, D., R.C. Lindsley, W. DeMuth, K. Rudd, S.A. Shinton, and R.R. Hardy. 2001. Resolution of three nonproliferative immature splenic B cell subsets reveals multiple selection points during peripheral B cell maturation. J. Immunol. 167:6834–6840. - PubMed

