Physiological and behavioral studies of spatial coding in the auditory cortex
- PMID: 17314017
- PMCID: PMC7116512
- DOI: 10.1016/j.heares.2007.01.001
Physiological and behavioral studies of spatial coding in the auditory cortex
Abstract
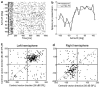
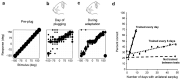
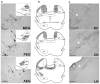
Despite extensive subcortical processing, the auditory cortex is believed to be essential for normal sound localization. However, we still have a poor understanding of how auditory spatial information is encoded in the cortex and of the relative contribution of different cortical areas to spatial hearing. We investigated the behavioral consequences of inactivating ferret primary auditory cortex (A1) on auditory localization by implanting a sustained release polymer containing the GABA(A) agonist muscimol bilaterally over A1. Silencing A1 led to a reversible deficit in the localization of brief noise bursts in both the horizontal and vertical planes. In other ferrets, large bilateral lesions of the auditory cortex, which extended beyond A1, produced more severe and persistent localization deficits. To investigate the processing of spatial information by high-frequency A1 neurons, we measured their binaural-level functions and used individualized virtual acoustic space stimuli to record their spatial receptive fields (SRFs) in anesthetized ferrets. We observed the existence of a continuum of response properties, with most neurons preferring contralateral sound locations. In many cases, the SRFs could be explained by a simple linear interaction between the acoustical properties of the head and external ears and the binaural frequency tuning of the neurons. Azimuth response profiles recorded in awake ferrets were very similar and further analysis suggested that the slopes of these functions and location-dependent variations in spike timing are the main information-bearing parameters. Studies of sensory plasticity can also provide valuable insights into the role of different brain areas and the way in which information is represented within them. For example, stimulus-specific training allows accurate auditory localization by adult ferrets to be relearned after manipulating binaural cues by occluding one ear. Reversible inactivation of A1 resulted in slower and less complete adaptation than in normal controls, whereas selective lesions of the descending cortico collicular pathway prevented any improvement in performance. These results reveal a role for auditory cortex in training-induced plasticity of auditory localization, which could be mediated by descending cortical pathways.
Figures





References
-
- Bajo VM, Nodal FR, Moore DR, King AJ. Role of auditory cortex and descending corticocollicular projections in adaptation to altered binaural cues by adult ferrets. Assoc Res Otolaryngol Abstr. 2006b;29:56.
-
- Beitel RE, Kaas JH. Effects of bilateral and unilateral ablation of auditory cortex in cats on the unconditioned head orienting response to acoustic stimuli. J Neurophysiol. 1993;70:351–369. - PubMed
-
- Belin P, Zatorre RJ. ‘What’, ‘where’ and ‘how’ in auditory cortex. Nature Neurosci. 2000;3:965–966. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

