Phosphomimetic mutation of Ser-187 of SNAP-25 increases both syntaxin binding and highly Ca2+-sensitive exocytosis
- PMID: 17325194
- PMCID: PMC2151612
- DOI: 10.1085/jgp.200609685
Phosphomimetic mutation of Ser-187 of SNAP-25 increases both syntaxin binding and highly Ca2+-sensitive exocytosis
Abstract
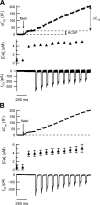
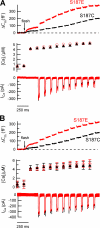
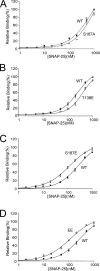
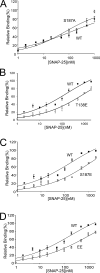
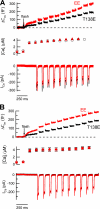
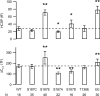
The phosphorylation targets that mediate the enhancement of exocytosis by PKC are unknown. PKC phosporylates the SNARE protein SNAP-25 at Ser-187. We expressed mutants of SNAP-25 using the Semliki Forest Virus system in bovine adrenal chromaffin cells and then directly measured the Ca2+ dependence of exocytosis using photorelease of caged Ca2+ together with patch-clamp capacitance measurements. A flash of UV light used to elevate [Ca2+](i) to several microM and release the highly Ca2+-sensitive pool (HCSP) of vesicles was followed by a train of depolarizing pulses to elicit exocytosis from the less Ca2+-sensitive readily releasable pool (RRP) of vesicles. Carbon fiber amperometry confirmed that the amount and kinetics of catecholamine release from individual granules were similar for the two phases of exocytosis. Mimicking PKC phosphorylation with expression of the S187E SNAP-25 mutant resulted in an approximately threefold increase in the HCSP, whereas the response to depolarization increased only 1.5-fold. The phosphomimetic S187D mutation resulted in an approximately 1.5-fold increase in the HCSP but a 30% smaller response to depolarization. In vitro binding assays with recombinant SNARE proteins were performed to examine shifts in protein-protein binding that may promote the highly Ca2+-sensitive state. The S187E mutant exhibited increased binding to syntaxin but decreased Ca2+-independent binding to synaptotagmin I. Mimicking phosphorylation of the putative PKA phosphorylation site of SNAP-25 with the T138E mutation decreased binding to both syntaxin and synaptotagmin I in vitro. Expressing the T138E/ S187E double mutant in chromaffin cells demonstrated that enhancing the size of the HCSP correlates with an increase in SNAP-25 binding to syntaxin in vitro, but not with Ca2+-independent binding of SNAP-25 to synaptotagmin I. Our results support the hypothesis that exocytosis triggered by lower Ca2+ concentrations (from the HCSP) occurs by different molecular mechanisms than exocytosis triggered by higher Ca2+ levels.
Figures


Similar articles
-
Ca2+-induced changes in SNAREs and synaptotagmin I correlate with triggered exocytosis from chromaffin cells: insights gleaned into the signal transduction using trypsin and botulinum toxins.J Cell Sci. 2002 Jul 1;115(Pt 13):2791-800. doi: 10.1242/jcs.115.13.2791. J Cell Sci. 2002. PMID: 12077369
-
Phosphorylation of SNAP-25 at Ser187 mediates enhancement of exocytosis by a phorbol ester in INS-1 cells.J Neurosci. 2008 Jan 2;28(1):21-30. doi: 10.1523/JNEUROSCI.2352-07.2008. J Neurosci. 2008. PMID: 18171919 Free PMC article.
-
Different effects on fast exocytosis induced by synaptotagmin 1 and 2 isoforms and abundance but not by phosphorylation.J Neurosci. 2006 Jan 11;26(2):632-43. doi: 10.1523/JNEUROSCI.2589-05.2006. J Neurosci. 2006. PMID: 16407561 Free PMC article.
-
Calcium signaling and exocytosis in adrenal chromaffin cells.Physiol Rev. 2006 Oct;86(4):1093-131. doi: 10.1152/physrev.00039.2005. Physiol Rev. 2006. PMID: 17015485 Review.
-
Rab3 and synaptotagmin proteins in the regulation of vesicle fusion and neurotransmitter release.Life Sci. 2022 Nov 15;309:120995. doi: 10.1016/j.lfs.2022.120995. Epub 2022 Sep 24. Life Sci. 2022. PMID: 36167148 Review.
Cited by
-
Phosphorylation within an autoinhibitory domain in endothelial nitric oxide synthase reduces the Ca(2+) concentrations required for calmodulin to bind and activate the enzyme.Biochemistry. 2008 Jul 15;47(28):7557-66. doi: 10.1021/bi8003186. Epub 2008 Jun 18. Biochemistry. 2008. PMID: 18558722 Free PMC article.
-
Automated targeting of cells to electrochemical electrodes using a surface chemistry approach for the measurement of quantal exocytosis.ACS Chem Neurosci. 2010 Jul 1;1(9):590-597. doi: 10.1021/cn1000183. ACS Chem Neurosci. 2010. PMID: 21113333 Free PMC article.
-
Electroporation followed by electrochemical measurement of quantal transmitter release from single cells using a patterned microelectrode.Lab Chip. 2013 Jun 7;13(11):2083-90. doi: 10.1039/c3lc41324a. Lab Chip. 2013. PMID: 23598689 Free PMC article.
-
Estimating amperometric spike parameters resulting from quantal exocytosis using curve fitting seeded by a matched-filter algorithm.J Neurosci Methods. 2019 Jan 1;311:360-368. doi: 10.1016/j.jneumeth.2018.09.018. Epub 2018 Sep 22. J Neurosci Methods. 2019. PMID: 30253199 Free PMC article.
-
A single amino acid mutation in SNAP-25 induces anxiety-related behavior in mouse.PLoS One. 2011;6(9):e25158. doi: 10.1371/journal.pone.0025158. Epub 2011 Sep 20. PLoS One. 2011. PMID: 21949876 Free PMC article.
References
-
- Ashery, U., A. Betz, T. Xu, N. Brose, and J. Rettig. 1999. An efficient method for infection of adrenal chromaffin cells using the Semliki Forest virus gene expression system. Eur. J. Cell Biol. 78:525–532. - PubMed
-
- Bai, J., C.T. Wang, D.A. Richards, M.B. Jackson, and E.R. Chapman. 2004. Fusion pore dynamics are regulated by synaptotagmin*t-SNARE interactions. Neuron. 41:929–942. - PubMed
-
- Craig, T.J., L.F. Ciufo, and A. Morgan. 2004. A protein-protein binding assay using coated microtitre plates: increased throughput, reproducibility and speed compared to bead-based assays. J. Biochem. Biophys. Methods. 60:49–60. - PubMed
-
- DiCiommo, D.P., and R. Bremner. 1998. Rapid, high level protein production using DNA-based Semliki Forest virus vectors. J. Biol. Chem. 273:18060–18066. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

