Arginase activity mediates reversible T cell hyporesponsiveness in human pregnancy
- PMID: 17330821
- PMCID: PMC2699382
- DOI: 10.1002/eji.200636542
Arginase activity mediates reversible T cell hyporesponsiveness in human pregnancy
Abstract
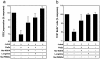
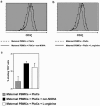
Complex regulation of T cell functions during pregnancy is required to ensure materno-fetal tolerance. Here we reveal a novel pathway for the temporary suppression of maternal T cell responses in uncomplicated human pregnancies. Our results show that arginase activity is significantly increased in the peripheral blood of pregnant women and remarkably high arginase activities are expressed in term placentae. High enzymatic activity results in high turnover of its substrate L-arginine and concomitant reduction of this amino acid in the microenvironment. Amino acid deprivation is emerging as a regulatory pathway of lymphocyte responses and we assessed the consequences of this enhanced arginase activity on T cell responses. Arginase-mediated L-arginine depletion induces down-regulation of CD3 zeta, the main signalling chain of the TCR, and functional T cell hyporesponsiveness. Importantly, this arginase-mediated T cell suppression was reversible, as inhibition of arginase activity or addition of exogenous L-arginine restored CD3 zeta chain expression and T cell proliferation. Thus, L-arginine metabolism constitutes a novel physiological mechanism contributing to the temporary suppression of the maternal immune response during human pregnancy.
Figures



References
-
- Medawar PB. Some immunological and endocrinological problems raised by the evolultion of viviparity in vertebrates. Symp. Soc. Exp. Biol. 1953;7:320–338.
-
- Hunt JS, Petroff MG, McIntire RH, Ober C. HLA-G and immune tolerance in pregnancy. FASEB J. 2005;19:681–693. - PubMed
-
- Petroff MG. Immune interactions at the maternal-fetal interface. J. Reprod. Immunol. 2005;68:1–13. - PubMed
-
- Tafuri A, Alferink J, Moller P, Hammerling GJ, Arnold B. T cell awareness of paternal alloantigens during pregnancy. Science. 1995;270:630–633. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

