Regulating cytoskeleton-based vesicle motility
- PMID: 17335816
- PMCID: PMC1974873
- DOI: 10.1016/j.febslet.2007.01.094
Regulating cytoskeleton-based vesicle motility
Abstract
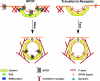
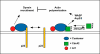
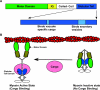
During vesicular transport, the assembly of the coat complexes and the selection of cargo proteins must be coordinated with the subsequent translocation of vesicles from the donor to an acceptor compartment. Here, we review recent progress toward uncovering the molecular mechanisms that connect transport vesicles to the protein machinery responsible for cytoskeleton-mediated motility. An emerging theme is that vesicle cargo proteins, either directly or through binding interactions with coat proteins, are able to influence cytoskeletal dynamics and motor protein function. Hence, a vesicle's cargo composition may help direct its intracellular motility and targeting.
Figures

Similar articles
-
Regulating the actin cytoskeleton during vesicular transport.Curr Opin Cell Biol. 2002 Aug;14(4):428-33. doi: 10.1016/s0955-0674(02)00349-6. Curr Opin Cell Biol. 2002. PMID: 12383793 Review.
-
Cargo takes control of endocytosis.Cell. 2006 Oct 6;127(1):35-7. doi: 10.1016/j.cell.2006.09.012. Cell. 2006. PMID: 17018271
-
Motoring around the Golgi.Nat Cell Biol. 2002 Oct;4(10):E236-42. doi: 10.1038/ncb1002-e236. Nat Cell Biol. 2002. PMID: 12360306 Review.
-
Coiled coils and SAH domains in cytoskeletal molecular motors.Biochem Soc Trans. 2011 Oct;39(5):1142-8. doi: 10.1042/BST0391142. Biochem Soc Trans. 2011. PMID: 21936779 Review.
-
Unconventional myosins: anchors in the membrane traffic relay.Traffic. 2000 Jan;1(1):11-8. doi: 10.1034/j.1600-0854.2000.010103.x. Traffic. 2000. PMID: 11208054 Review.
Cited by
-
Spatial modeling of vesicle transport and the cytoskeleton: the challenge of hitting the right road.PLoS One. 2012;7(1):e29645. doi: 10.1371/journal.pone.0029645. Epub 2012 Jan 12. PLoS One. 2012. PMID: 22253752 Free PMC article.
-
Galectin-9 interacts with Vamp-3 to regulate cytokine secretion in dendritic cells.Cell Mol Life Sci. 2023 Sep 27;80(10):306. doi: 10.1007/s00018-023-04954-x. Cell Mol Life Sci. 2023. PMID: 37755527 Free PMC article.
-
Identification of Transferrin Receptor 1 (TfR1) Overexpressed in Lung Cancer Cells, and Internalization of Magnetic Au-CoFe2O4 Core-Shell Nanoparticles Functionalized with Its Ligand in a Cellular Model of Small Cell Lung Cancer (SCLC).Pharmaceutics. 2022 Aug 17;14(8):1715. doi: 10.3390/pharmaceutics14081715. Pharmaceutics. 2022. PMID: 36015341 Free PMC article.
-
Actin acting at the Golgi.Histochem Cell Biol. 2013 Sep;140(3):347-60. doi: 10.1007/s00418-013-1115-8. Epub 2013 Jun 27. Histochem Cell Biol. 2013. PMID: 23807268 Review.
-
The role of actin remodeling in the trafficking of intracellular vesicles, transporters, and channels: focusing on aquaporin-2.Pflugers Arch. 2008 Jul;456(4):737-45. doi: 10.1007/s00424-007-0404-2. Epub 2007 Dec 8. Pflugers Arch. 2008. PMID: 18066585 Review.
References
-
- Caviston JP, Holzbaur EL. Microtubule motors at the intersection of trafficking and transport. Trends Cell Biol. 2006;16:530–7. - PubMed
-
- Soldati T, Schliwa M. Powering membrane traffic in endocytosis and recycling. Nat Rev Mol Cell Biol. 2006;7:897–908. - PubMed
-
- Egea G, Lazaro-Dieguez F, Vilella M. Actin dynamics at the Golgi complex in mammalian cells. Curr Opin Cell Biol. 2006;18:168–178. - PubMed
-
- Orci L, Stamnes M, Ravazzola M, Amherdt M, Perrelet A, Sollner TH, Rothman JE. Bidirectional transport by distinct populations of COPI-coated vesicles. Cell. 1997;90:335–49. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

