Incorporation of human immunodeficiency virus type 1 reverse transcriptase into virus-like particles
- PMID: 17344306
- PMCID: PMC1900205
- DOI: 10.1128/JVI.01796-06
Incorporation of human immunodeficiency virus type 1 reverse transcriptase into virus-like particles
Abstract
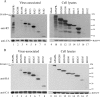
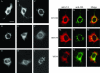
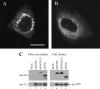
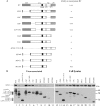
We demonstrate that a genetically engineered human immunodeficiency virus type 1 (HIV-1) reverse transcriptase (RT) composed mainly of p66 or p51 subunits can be incorporated into virus-like particles (VLPs) when coexpressed with HIV-1 Pr55(gag). VLP-associated RT exhibited a detergent-resistant association with immature cores during sucrose gradient equilibrium centrifugation, suggesting that RT is incorporated into VLPs. However, RT that retains downstream integrase (IN) is severely inhibited in terms of incorporation into VLPs. Results from immunofluorescence tests reveal that RT-IN is primarily localized at the perinuclear area and exhibits poor colocalization with Gag. IN removal leads to a redistribution of RT throughout the cytoplasm and improved RT incorporation into VLPs. Similar results were observed for RT-IN in which alanine was substituted for 186-Lys-Arg-Lys-188 residues of the IN putative nuclear localization signal, suggesting that IN karyophilic properties may partly account for the inhibitory effect of IN on RT incorporation. Although the membrane-binding capacity of RT was markedly reduced compared to that of wild-type Gag or Gag-Pol, the correlation of membrane-binding ability with particle incorporation efficiency was incomplete. Furthermore, we observed that membrane-binding-defective myristylation-minus RT can be packaged into VLPs at the same level as its normal myristylated counterpart. This suggests that the incorporation of RT into VLPs is independent of membrane affinity but very dependent on RT-Gag interaction. Results from a genetic analysis suggest that the Gag-interacting regions of RT mainly reside in the thumb subdomain and that the RT-binding domains of Gag are located in the matrix (MA) and p6 regions.
Figures



Similar articles
-
A virus-like particle assembly system for probing the HIV-1 Gag-Pol dimerization domain: supporting evidence for reverse transcriptase involvement in protease activation by influencing Gag-Pol/Gag-Pol interaction.J Virol. 2025 Apr 15;99(4):e0223624. doi: 10.1128/jvi.02236-24. Epub 2025 Mar 12. J Virol. 2025. PMID: 40071921 Free PMC article.
-
Characterization of human immunodeficiency virus type 1 Pr160 gag-pol mutants with truncations downstream of the protease domain.Virology. 2004 Nov 10;329(1):180-8. doi: 10.1016/j.virol.2004.08.010. Virology. 2004. PMID: 15476885
-
Coding sequences upstream of the human immunodeficiency virus type 1 reverse transcriptase domain in Gag-Pol are not essential for incorporation of the Pr160(gag-pol) into virus particles.J Virol. 2002 Apr;76(7):3221-31. doi: 10.1128/jvi.76.7.3221-3231.2002. J Virol. 2002. PMID: 11884546 Free PMC article.
-
Incorporation of pol into human immunodeficiency virus type 1 Gag virus-like particles occurs independently of the upstream Gag domain in Gag-pol.J Virol. 2004 Jan;78(2):1042-9. doi: 10.1128/jvi.78.2.1042-1049.2004. J Virol. 2004. PMID: 14694138 Free PMC article.
-
The HIV-1 assembly machine.AIDS. 2001;15 Suppl 5:S13-20. doi: 10.1097/00002030-200100005-00003. AIDS. 2001. PMID: 11816161 Review. No abstract available.
Cited by
-
Placement of leucine zipper motifs at the carboxyl terminus of HIV-1 protease significantly reduces virion production.PLoS One. 2012;7(3):e32845. doi: 10.1371/journal.pone.0032845. Epub 2012 Mar 1. PLoS One. 2012. PMID: 22396796 Free PMC article.
-
HIV Genome-Wide Protein Associations: a Review of 30 Years of Research.Microbiol Mol Biol Rev. 2016 Jun 29;80(3):679-731. doi: 10.1128/MMBR.00065-15. Print 2016 Sep. Microbiol Mol Biol Rev. 2016. PMID: 27357278 Free PMC article.
-
A single amino acid substitution in HIV-1 reverse transcriptase significantly reduces virion release.J Virol. 2010 Jan;84(2):976-82. doi: 10.1128/JVI.01532-09. Epub 2009 Nov 4. J Virol. 2010. PMID: 19889767 Free PMC article.
-
A virus-like particle assembly system for probing the HIV-1 Gag-Pol dimerization domain: supporting evidence for reverse transcriptase involvement in protease activation by influencing Gag-Pol/Gag-Pol interaction.J Virol. 2025 Apr 15;99(4):e0223624. doi: 10.1128/jvi.02236-24. Epub 2025 Mar 12. J Virol. 2025. PMID: 40071921 Free PMC article.
-
The y271 and i274 amino acids in reverse transcriptase of human immunodeficiency virus-1 are critical to protein stability.PLoS One. 2009 Jul 3;4(7):e6108. doi: 10.1371/journal.pone.0006108. PLoS One. 2009. PMID: 19578544 Free PMC article.
References
-
- Abdurahman, S., S. Hoglund, L. Goobar-Larsson, and A. Vahlne. 2004. Selected amino acid substitutions in the C-terminal region of human immunodeficiency virus type 1 capsid protein affect virus assembly and release. J. Gen. Virol. 85:2903-2913. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

