Additive protective effects of the addition of lactic acid and adrenaline on excitability and force in isolated rat skeletal muscle depressed by elevated extracellular K+
- PMID: 17347268
- PMCID: PMC2075200
- DOI: 10.1113/jphysiol.2007.129049
Additive protective effects of the addition of lactic acid and adrenaline on excitability and force in isolated rat skeletal muscle depressed by elevated extracellular K+
Abstract
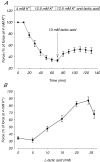
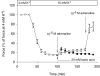
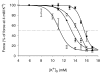
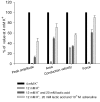
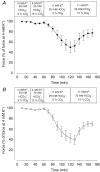
During strenuous exercise, extracellular K(+) ([K(+)](o)) is increased, which potentially can reduce muscle excitability and force production. In addition, exercise leads to accumulation of lactate and H(+) and increased levels of circulating catecholamines. Individually, reduced pH and increased catecholamines have been shown to counteract the depressing effect of elevated K(+). This study examines (i) whether the effects of addition of lactic acid and adrenaline on the excitability of isolated muscles are caused by separate mechanisms and are additive and (ii) whether the effect of adding lactic acid or increasing CO(2) is related to a reduction of intra- or extracellular pH. Rat soleus muscles were incubated at a [K(+)](o) of 15 mM, which reduced tetanic force by 85%. Subsequent addition of 20 mM lactic acid or 10(-5) M adrenaline led to a small recovery of force, but when added together induced an almost complete force recovery. Compound action potentials showed that the force recovery was associated with recovery of muscle excitability. The improved excitability after addition of adrenaline was associated with increased Na(+)-K(+) pump activity resulting in hyperpolarization and an increase in the chemical Na(+) gradient. In contrast, addition of lactic acid had no effect on the membrane potential or the Na(+)-K(+) pump activity, but most likely increased excitability via a reduction in intracellular pH. It is concluded that the protective effects of acidosis and adrenaline on muscle excitability and force took place via different mechanisms and were additive. The results suggest that circulating catecholamines and development of acidosis during exercise may improve the tolerance of muscles to elevated [K(+)](o).
Figures

Comment in
-
Combating muscle fatigue: extracellular lactic acidosis and catecholamines.J Physiol. 2007 Jun 1;581(Pt 2):419. doi: 10.1113/jphysiol.2007.132209. Epub 2007 Mar 22. J Physiol. 2007. PMID: 17379626 Free PMC article. No abstract available.
References
-
- Albuquerque EX, Thesleff S. A comparative study of membrane properties of innervated and chronically denervated fast and slow skeletal muscles of the rat. Acta Physiol Scand. 1968;73:471–480. - PubMed
-
- Andersen SL, Clausen T. Calcitonin gene-related peptide stimulates active Na+-K+ transport in rat soleus muscle. Am J Physiol. 1993;268:C1528–C1536. - PubMed
-
- Bouclin R, Charbonneau E, Renaud JM. Na+ and K+ effect on contractility of frog sartorius muscle: implication for the mechanism of fatigue. Am J Physiol. 1995;268:C1528–C1536. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

