Predominantly buried residues in the response regulator Spo0F influence specific sensor kinase recognition
- PMID: 17350627
- PMCID: PMC1987376
- DOI: 10.1016/j.febslet.2007.02.061
Predominantly buried residues in the response regulator Spo0F influence specific sensor kinase recognition
Abstract
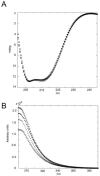
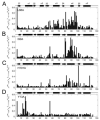
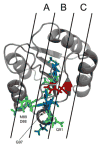
Several alanine mutations in the response regulator Spo0F induce hypersporulation in Bacillus subtilis. L66A, I90A and H101A mutants are purported to be involved in contacts stabilizing the orientation of the alpha4-helix and hence the beta4-alpha4 kinase recognition loop. Y13A is thought to affect the orientation of the alpha1-helix and consequently phosphatase action. Using comparative NMR chemical shift analyses for these mutants, we have confirmed these suppositions and isolated residues in Spo0F critical in sensor kinases discrimination. In addition, we discuss how buried residues and intra-protein communication networks contribute to precise molecular recognition by ensuring that the correct surface is presented.
Figures
Similar articles
-
Alanine mutants of the Spo0F response regulator modifying specificity for sensor kinases in sporulation initiation.Mol Microbiol. 1999 Jul;33(2):389-95. doi: 10.1046/j.1365-2958.1999.01481.x. Mol Microbiol. 1999. PMID: 10411754
-
Characterization of interactions between a two-component response regulator, Spo0F, and its phosphatase, RapB.Biochemistry. 1998 Nov 24;37(47):16538-45. doi: 10.1021/bi981340o. Biochemistry. 1998. PMID: 9843420
-
Molecular recognition in signal transduction: the interaction surfaces of the Spo0F response regulator with its cognate phosphorelay proteins revealed by alanine scanning mutagenesis.J Mol Biol. 1997 Sep 19;272(2):200-12. doi: 10.1006/jmbi.1997.1226. J Mol Biol. 1997. PMID: 9299348
-
Overview of protein phosphorylation in bacteria with a main focus on unusual protein kinases in Bacillus subtilis.Res Microbiol. 2021 Nov-Dec;172(7-8):103871. doi: 10.1016/j.resmic.2021.103871. Epub 2021 Sep 6. Res Microbiol. 2021. PMID: 34500011 Review.
-
[Sporulation in Bacillus subtilis: signal transduction at the initiation of sporulation].Tanpakushitsu Kakusan Koso. 1995 Jun;40(8):976-85. Tanpakushitsu Kakusan Koso. 1995. PMID: 7610262 Review. Japanese. No abstract available.
Cited by
-
Identification of Functional Spo0A Residues Critical for Sporulation in Clostridioides difficile.J Mol Biol. 2022 Jul 15;434(13):167641. doi: 10.1016/j.jmb.2022.167641. Epub 2022 May 18. J Mol Biol. 2022. PMID: 35597553 Free PMC article.
-
Long range dynamic effects of point-mutations trap a response regulator in an active conformation.FEBS Lett. 2010 Oct 8;584(19):4203-7. doi: 10.1016/j.febslet.2010.08.051. Epub 2010 Sep 7. FEBS Lett. 2010. PMID: 20828564 Free PMC article.
-
Toward rationally redesigning bacterial two-component signaling systems using coevolutionary information.Proc Natl Acad Sci U S A. 2014 Feb 4;111(5):E563-71. doi: 10.1073/pnas.1323734111. Epub 2014 Jan 21. Proc Natl Acad Sci U S A. 2014. PMID: 24449878 Free PMC article.
-
High-resolution protein complexes from integrating genomic information with molecular simulation.Proc Natl Acad Sci U S A. 2009 Dec 29;106(52):22124-9. doi: 10.1073/pnas.0912100106. Epub 2009 Dec 17. Proc Natl Acad Sci U S A. 2009. PMID: 20018738 Free PMC article.
-
Structure of the Acinetobacter baumannii PmrA receiver domain and insights into clinical mutants affecting DNA binding and promoting colistin resistance.J Biochem. 2022 Jan 7;170(6):787-800. doi: 10.1093/jb/mvab102. J Biochem. 2022. PMID: 34585233 Free PMC article.
References
-
- Burbulys D, Trach KA, Hoch JA. Initiation of sporulation in B. subtilis is controlled by a multicomponent phosphorelay. Cell. 1991;64:545–52. - PubMed
-
- Hoch JA. Regulation of the onset of the stationary phase and sporulation in Bacillus subtilis. Adv Microb Physiol. 1993;35:111–33. - PubMed
-
- Hoch JA. Regulation of the phosphorelay and the initiation of sporulation in Bacillus subtilis. Annu Rev Microbiol. 1993;47:441–65. - PubMed
-
- Perego M. A new family of aspartyl phosphate phosphatases targeting the sporulation transcription factor Spo0A of Bacillus subtilis. Mol Microbiol. 2001;42:133–43. - PubMed
-
- Jiang M, Tzeng YL, Feher VA, Perego M, Hoch JA. Alanine mutants of the Spo0F response regulator modifying specificity for sensor kinases in sporulation initiation. Mol Microbiol. 1999;33:389–95. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

