Role of matrix metalloproteinases in the acquisition and reconsolidation of cocaine-induced conditioned place preference
- PMID: 17353546
- PMCID: PMC1838561
- DOI: 10.1101/lm.476207
Role of matrix metalloproteinases in the acquisition and reconsolidation of cocaine-induced conditioned place preference
Abstract
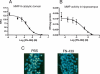
Persistent drug seeking/taking behavior involves the consolidation of memory. With each drug use, the memory may be reactivated and reconsolidated to maintain the original memory. During reactivation, the memory may become labile and susceptible to disruption; thus, molecules involved in plasticity should influence acquisition and/or reconsolidation. Recently, matrix metalloproteinases (MMPs) have been shown to influence neuronal plasticity, presumably by their regulation of extracellular matrix (ECM) molecules involved in synaptic reorganization during learning. We hypothesized that inhibition of MMP activity would impair the acquisition and/or reconsolidation of cocaine-conditioned place preference (CPP) in rats. Intracerebral ventricular (i.c.v.) microinjection of a broad spectrum MMP inhibitor, FN-439, prior to cocaine training suppressed acquisition of CPP and attenuated cocaine-primed reinstatement in extinguished animals. In a separate experiment, the cocaine memory was reactivated on two consecutive days with a cocaine priming injection. On these two days, artificial cerebral spinal fluid (aCSF) or FN-439 was administered either 30 min prior to or 1 min after cocaine-primed reinstatement sessions. Infusion of FN-439 partially impaired retrieval of the cocaine-associated context when given 30 min prior to cocaine. In both groups, however, FN-439 suppressed reinstatement compared with controls on the third consecutive test for cocaine-primed reinstatement, when no FN-439 was given. Control experiments demonstrated that two injections of FN-439 + cocaine given in the home cage, or of FN-439 + saline priming injections in the CPP chambers did not disrupt subsequent cocaine-primed reinstatement. These results show for the first time that (1) MMPs play a critical role in acquisition and reconsolidation of cocaine-induced CPP, and (2) rats demonstrate apparent disruption of reconsolidation by an MMP inhibitor after extinction and while they are under the influence of cocaine during reinstatement.
Figures






References
-
- Bahar A., Dorfman N., Dudai Y. Amygdalar circuits required for either consolidation or extinction of taste aversion memory are not required for reconsolidation. Eur. J. Neurosci. 2004;19:1115–1118. - PubMed
-
- Bernardi R.E., Lattal K.M., Berger S.P. Postretrieval propranolol disrupts a cocaine conditioned place preference. Neuroreport. 2006;17:1443–1447. - PubMed
-
- Brenz Verca M.S., Widmer D.A., Wagner G.C., Dreyer J. Cocaine-induced expression of the tetraspanin CD81 and its relation to hypothalamic function. Mol. Cell. Neurosci. 2001;17:303–316. - PubMed
-
- Conant K., St Hillaire C., Nagase H., Visse R., Gary D., Haughey N., Anderson C., Turchan J., Nath A. Matrix metalloproteinase 1 interacts with neuronal integrins and stimulates dephosphorylation of Akt. J. Biol. Chem. 2004;279:8056–8062. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
