At diabetes-like concentration, glucose down-regulates the placental serotonin transport system in a cell-cycle-dependent manner
- PMID: 17355243
- PMCID: PMC3042026
- DOI: 10.1111/j.1471-4159.2007.04469.x
At diabetes-like concentration, glucose down-regulates the placental serotonin transport system in a cell-cycle-dependent manner
Retraction in
-
Retraction.J Neurochem. 2020 Apr;153(1):138. doi: 10.1111/jnc.14931. Epub 2020 Feb 17. J Neurochem. 2020. PMID: 32202334 Free PMC article.
Abstract
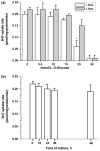
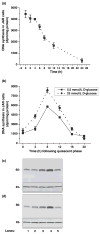
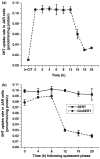
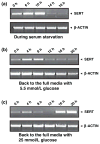
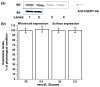
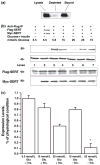
Serotonin [5-hydroxytryptamine (5HT)] is a vasoconstrictor that also acts as a developmental signal early in embryogenesis. The 5HT transporter (SERT) on the membranes of the placental trophoblast cells controls 5HT levels in the maternal bloodstream to maintain stable transplacental blood flow and simultaneously provide 5HT to the embryo. The 5HT uptake rate of placental SERT is important for both the mother and the developing embryo. The impact of glucose on the placental SERT system during diabetic pregnancy is not known. The present in vitro study investigated this important issue in human placental choriocarcinoma (JAR) cells that were cultured for 24-96 h in a medium containing either 5.5 (physiologic concentration) or 25 mmol/L D-glucose (diabetic-like concentration). The 5HT uptake rates of the cultured cells were not altered at exogenous D-glucose concentrations in the range of 5.5-15 mmol/L, but were decreased significantly at a diabetic-like concentration (>or=25 mmol/L). To understand better the role of glucose on the placental 5HT system, we first characterized SERT in JAR cells at different cell-cycle phases and then determined the expression levels of SERT on the plasma membrane and in the intracellular pools of JAR cells at the late-S and G2 phases, where the uptake rates were decreased 73% under diabetic-like glucose concentrations. Finally, the importance of self-association of SERT molecules was examined. In JAR cells co-expressing Flag- and myc-tagged SERT, myc-antibody precipitated 70% of Flag-SERT, indicating that a large percentage of SERT proteins exist as oligomers in situ. Under diabetic conditions, myc-antibody no longer precipitated Flag-SERT, suggesting a disruption in the aggregation of SERT molecules. Therefore, we propose that under uncontrolled diabetic conditions, glucose down-regulates 5HT uptake rates of placental SERT by interfering with its functional expression in a cell-cycle-dependent manner.
Figures

Similar articles
-
Uptake of 1-methyl-4-phenylpyridinium (MPP+) by the JAR human placental choriocarcinoma cell line: comparison with 5-hydroxytryptamine.Placenta. 2003 Apr;24(4):361-9. doi: 10.1053/plac.2002.0917. Placenta. 2003. PMID: 12657510
-
Serotonin transamidates Rab4 and facilitates its binding to the C terminus of serotonin transporter.J Biol Chem. 2008 Apr 4;283(14):9388-98. doi: 10.1074/jbc.M706367200. Epub 2008 Jan 28. J Biol Chem. 2008. Retraction in: J Biol Chem. 2019 Jun 14;294(24):9658. doi: 10.1074/jbc.W119.009467. PMID: 18227069 Retracted.
-
The cellular distribution of serotonin transporter is impeded on serotonin-altered vimentin network.PLoS One. 2009;4(3):e4730. doi: 10.1371/journal.pone.0004730. Epub 2009 Mar 9. PLoS One. 2009. Retraction in: PLoS One. 2019 Feb 1;14(2):e0211966. doi: 10.1371/journal.pone.0211966. PMID: 19270731 Free PMC article. Retracted.
-
Molecular mechanisms of SERT in platelets: regulation of plasma serotonin levels.Mol Interv. 2010 Aug;10(4):231-41. doi: 10.1124/mi.10.4.6. Mol Interv. 2010. PMID: 20729489 Free PMC article. Review.
-
Going with the flow: trafficking-dependent and -independent regulation of serotonin transport.Traffic. 2008 Sep;9(9):1393-402. doi: 10.1111/j.1600-0854.2008.00757.x. Epub 2008 Apr 28. Traffic. 2008. PMID: 18445122 Free PMC article. Review.
Cited by
-
Epigenetic adaptation of the placental serotonin transporter gene (SLC6A4) to gestational diabetes mellitus.PLoS One. 2017 Jun 26;12(6):e0179934. doi: 10.1371/journal.pone.0179934. eCollection 2017. PLoS One. 2017. PMID: 28650965 Free PMC article.
-
GDM-associated insulin deficiency hinders the dissociation of SERT from ERp44 and down-regulates placental 5-HT uptake.Proc Natl Acad Sci U S A. 2014 Dec 30;111(52):E5697-705. doi: 10.1073/pnas.1416675112. Epub 2014 Dec 15. Proc Natl Acad Sci U S A. 2014. PMID: 25512553 Free PMC article. Clinical Trial.
-
Association with serotonin transporter enables the phosphorylation of insulin receptor in placenta.Curr Top Biochem Res. 2019;20:65-78. Curr Top Biochem Res. 2019. PMID: 38327526 Free PMC article.
-
Drug transporters in the human blood-placental barrier.Br J Pharmacol. 2009 Oct;158(3):665-78. doi: 10.1111/j.1476-5381.2009.00336.x. Epub 2009 Sep 25. Br J Pharmacol. 2009. PMID: 19788499 Free PMC article. Review.
References
-
- Balkovetz DF, Tiruppathi C, Leibach FH, Mahesh VB, Ganapathy V. Evidence for an imipramine-sensitive serotonin transporter in human placental brush-border membranes. J Biol Chem. 1989;264:2195–2198. - PubMed
-
- Bengel D, Murphy DL, Andrews AM, Wichems CH, Feltner D, Heils A, Mossner R, Westphal H, Lesch K. Altered brain serotonin homeostasis and locomotor insensitivity to 3,4-methylenedioxymethamphetamine (“Ecstasy”) in serotonin transporter-deficient mice. Mol Pharmacol. 1998;53:649–655. - PubMed
-
- Blakely RD, Berson HE, Fremeau RT, Jr, Caron MC, Peek MM, Priace HK, Bradley CC. Cloning and expression of a functional serotonin transporter from rat brain. Nature. 1991;354:66–70. - PubMed
-
- Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature. 2001;414:813–820. - PubMed
-
- Cases O, Vitalis T, Seif I, De Maeyer E, Sotelo C, Gaspar P. Lack of barrels in the somatosensory cortex of monoamine oxidase A-deficient mice: role of a serotonin excess during the critical period. Neuron. 1996;16:297–307. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

