Inhibition of ADAM17 reduces hypoxia-induced brain tumor cell invasiveness
- PMID: 17355261
- PMCID: PMC11158419
- DOI: 10.1111/j.1349-7006.2007.00440.x
Inhibition of ADAM17 reduces hypoxia-induced brain tumor cell invasiveness
Abstract
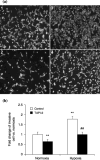
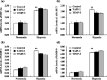
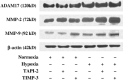
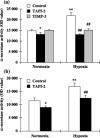
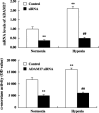
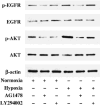
The membrane-anchored metalloproteinase tumor necrosis factor-alpha-converting enzyme (TACE/a disintegrin and metalloproteinase [ADAM] 17) is key in proteolytic ectodomain shedding of several membrane-bound growth factors, cytokines and receptors. The expression and activity of ADAM17 increases under some pathological conditions including stroke, and promotes neural progenitor cell migration and contributes to stroke-induced neurogenesis. Hypoxia initiates cellular invasive processes that occur under both physiological and pathological conditions such as invasion and metastasis of some tumors. In the present study, we sought to elucidate whether ADAM17 contributes to brain tumor invasion. To this end, we examined the role of ADAM17 in the invasiveness of two different brain tumor cell lines, 9L rat gliosarcoma and U87 human glioma, under normoxic and hypoxic conditions. Additionally, we tested the effects of ADAM17 suppression on in vitro tumor cell invasion by means of ADAM17 proteolytic inhibitors and specific small interfering RNA. We found that tumor cells upregulated ADAM17 expression under hypoxia, and that ADAM17 activity correlated with increased tumor cell invasion. Conversely, suppression of ADAM17 proteolysis decreased invasiveness induced by hypoxia in 9L and U87 cells. Furthermore, the contribution of ADAM17 to tumor invasion was independent of matrix metalloproteinase (MMP)-2 and MMP-9 activity. ADAM17 was also found to activate the epidermal growth factor/phosphoinositide-3 kinase/serine/threonine kinase signal transduction pathway. Our data suggest that hypoxia-induced ADAM17 contributes to glioma cell invasiveness through activation of the EGFR signal pathway.
Figures






Similar articles
-
ADAM17 promotes breast cancer cell malignant phenotype through EGFR-PI3K-AKT activation.Cancer Biol Ther. 2009 Jun;8(11):1045-54. doi: 10.4161/cbt.8.11.8539. Epub 2009 Jun 25. Cancer Biol Ther. 2009. PMID: 19395875 Free PMC article.
-
ADAM17 targets MMP-2 and MMP-9 via EGFR-MEK-ERK pathway activation to promote prostate cancer cell invasion.Int J Oncol. 2012 May;40(5):1714-24. doi: 10.3892/ijo.2011.1320. Epub 2011 Dec 23. Int J Oncol. 2012. PMID: 22200661
-
ADAM17 promotes glioma cell malignant phenotype.Mol Carcinog. 2012 Feb;51(2):150-64. doi: 10.1002/mc.20772. Epub 2011 Apr 7. Mol Carcinog. 2012. PMID: 21480393 Free PMC article.
-
The role of ADAM17 in metabolic inflammation.Atherosclerosis. 2013 May;228(1):12-7. doi: 10.1016/j.atherosclerosis.2013.01.024. Epub 2013 Jan 25. Atherosclerosis. 2013. PMID: 23384719 Review.
-
Contribution of ADAM17 and related ADAMs in cardiovascular diseases.Cell Mol Life Sci. 2021 May;78(9):4161-4187. doi: 10.1007/s00018-021-03779-w. Epub 2021 Feb 11. Cell Mol Life Sci. 2021. PMID: 33575814 Free PMC article. Review.
Cited by
-
Metalloproteinases ADAM10 and ADAM17 Mediate Migration and Differentiation in Glioblastoma Sphere-Forming Cells.Mol Neurobiol. 2017 Jul;54(5):3893-3905. doi: 10.1007/s12035-016-0053-6. Epub 2016 Aug 19. Mol Neurobiol. 2017. PMID: 27541285 Free PMC article.
-
Expression of a-disintegrin and metalloproteinase 10 correlates with grade of malignancy in human glioma.Oncol Lett. 2015 May;9(5):2157-2162. doi: 10.3892/ol.2015.2993. Epub 2015 Feb 27. Oncol Lett. 2015. PMID: 26137031 Free PMC article.
-
A disintegrin and metalloprotease 17 promotes microglial cell survival via epidermal growth factor receptor signalling following spinal cord injury.Mol Med Rep. 2015 Jul;12(1):63-70. doi: 10.3892/mmr.2015.3395. Epub 2015 Feb 27. Mol Med Rep. 2015. Retraction in: Mol Med Rep. 2025 Feb;31(2):49. doi: 10.3892/mmr.2024.13414. PMID: 25738567 Free PMC article. Retracted.
-
Tumorigenicity of cortical astrocyte cell line induced by the protease ADAM17.Cancer Sci. 2009 Sep;100(9):1597-604. doi: 10.1111/j.1349-7006.2009.01221.x. Epub 2009 May 18. Cancer Sci. 2009. PMID: 19515085 Free PMC article.
-
ADAM17 promotes breast cancer cell malignant phenotype through EGFR-PI3K-AKT activation.Cancer Biol Ther. 2009 Jun;8(11):1045-54. doi: 10.4161/cbt.8.11.8539. Epub 2009 Jun 25. Cancer Biol Ther. 2009. PMID: 19395875 Free PMC article.
References
-
- Black RA. Tumor necrosis factor‐α converting enzyme. Int J Biochem Cell Biol 2002; 34: 1–5. - PubMed
-
- Black RA, Rauch CT, Kozlosky CJ et al. A metalloproteinase disintegrin that releases tumour‐necrosis factor‐α from cells. Nature 1997; 385: 729–33. - PubMed
-
- Buxbaum JD, Liu KN, Luo Y et al. Evidence that tumor necrosis factor alpha converting enzyme is involved in regulated alpha‐secretase cleavage of the Alzheimer amyloid protein precursor. J Biol Chem 1998; 273: 27 765–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

