Estrogen and resveratrol regulate Rac and Cdc42 signaling to the actin cytoskeleton of metastatic breast cancer cells
- PMID: 17356711
- PMCID: PMC1813930
- DOI: 10.1593/neo.06778
Estrogen and resveratrol regulate Rac and Cdc42 signaling to the actin cytoskeleton of metastatic breast cancer cells
Abstract
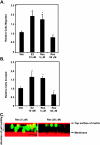
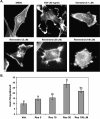
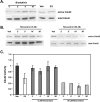
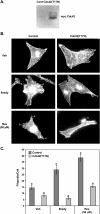
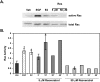
Estrogen and structurally related molecules play critical roles in breast cancer. We reported that resveratrol (50 microM), an estrogen-like phytosterol from grapes, acts in an antiestrogenic manner in breast cancer cells to reduce cell migration and to induce a global and sustained extension of actin structures called filopodia. Herein, we report that resveratrol-induced filopodia formation is time-dependent and concentration-dependent. In contrast to resveratrol at 50 microM, resveratrol at 5 microM acts in a manner similar to estrogen by increasing lamellipodia, as well as cell migration and invasion. Because Rho GTPases regulate the extension of actin structures, we investigated a role for Rac and Cdc42 in estrogen and resveratrol signaling. Our results demonstrate that 50 microM resveratrol decreases Rac and Cdc42 activity, whereas estrogen and 5 microM resveratrol increase Rac activity in breast cancer cells. MDA-MB-231 cells expressing dominant-negative Cdc42 or dominant-negative Rac retain filopodia response to 50 microM resveratrol. Lamellipodia response to 5 microM resveratrol, estrogen, or epidermal growth factor is inhibited in cells expressing dominant-negative Rac, indicating that Rac regulates estrogen and resveratrol (5 microM) signaling to the actin cytoskeleton. These results indicate that signaling to the actin cytoskeleton by low and high concentrations of resveratrol may be differentially regulated by Rac and Cdc42.
Figures



Similar articles
-
Resveratrol and estradiol exert disparate effects on cell migration, cell surface actin structures, and focal adhesion assembly in MDA-MB-231 human breast cancer cells.Neoplasia. 2005 Feb;7(2):128-40. doi: 10.1593/neo.04346. Neoplasia. 2005. PMID: 15802018 Free PMC article.
-
The TRE17 oncogene encodes a component of a novel effector pathway for Rho GTPases Cdc42 and Rac1 and stimulates actin remodeling.Mol Cell Biol. 2003 Mar;23(6):2151-61. doi: 10.1128/MCB.23.6.2151-2161.2003. Mol Cell Biol. 2003. PMID: 12612085 Free PMC article.
-
Involvement of Cdc42 and Rac small G proteins in invadopodia formation of RPMI7951 cells.Genes Cells. 2003 Dec;8(12):1019-27. doi: 10.1111/j.1365-2443.2003.00695.x. Genes Cells. 2003. PMID: 14750956
-
Regulation of phosphorylation pathways by p21 GTPases. The p21 Ras-related Rho subfamily and its role in phosphorylation signalling pathways.Eur J Biochem. 1996 Dec 1;242(2):171-85. doi: 10.1111/j.1432-1033.1996.0171r.x. Eur J Biochem. 1996. PMID: 8973630 Review.
-
Rho, Rac and Cdc42 GTPases regulate the organization of the actin cytoskeleton.Curr Opin Cell Biol. 1997 Feb;9(1):86-92. doi: 10.1016/s0955-0674(97)80156-1. Curr Opin Cell Biol. 1997. PMID: 9013670 Review.
Cited by
-
A quantitative proteomics analysis of MCF7 breast cancer stem and progenitor cell populations.Proteomics. 2015 Nov;15(22):3772-83. doi: 10.1002/pmic.201500002. Epub 2015 Oct 7. Proteomics. 2015. PMID: 26332018 Free PMC article.
-
Coordinate regulation of estrogen-mediated fibronectin matrix assembly and epidermal growth factor receptor transactivation by the G protein-coupled receptor, GPR30.Mol Endocrinol. 2009 Jul;23(7):1052-64. doi: 10.1210/me.2008-0262. Epub 2009 Apr 2. Mol Endocrinol. 2009. PMID: 19342448 Free PMC article.
-
Rapid actions of plasma membrane estrogen receptors regulate motility of mouse embryonic stem cells through a profilin-1/cofilin-1-directed kinase signaling pathway.Mol Endocrinol. 2012 Aug;26(8):1291-303. doi: 10.1210/me.2012-1002. Epub 2012 Jun 25. Mol Endocrinol. 2012. PMID: 22734041 Free PMC article.
-
Target proteomic profiling of frozen pancreatic CD24+ adenocarcinoma tissues by immuno-laser capture microdissection and nano-LC-MS/MS.J Proteome Res. 2013 Jun 7;12(6):2791-804. doi: 10.1021/pr400139c. Epub 2013 May 29. J Proteome Res. 2013. PMID: 23679566 Free PMC article.
-
Multifaceted approach to resveratrol bioactivity: Focus on antioxidant action, cell signaling and safety.Oxid Med Cell Longev. 2010 Mar-Apr;3(2):86-100. doi: 10.4161/oxim.3.2.11147. Oxid Med Cell Longev. 2010. PMID: 20716933 Free PMC article. Review.
References
-
- Acconcia F, Barnes CJ, Kumar R. Estrogen and tamoxifen induce cytoskeletal remodeling and migration in endometrial cancer cells. Endocrinology. 2006;147:1203–1212. - PubMed
-
- Song RX, Santen RJ. Membrane initiated estrogen signaling in breast cancer. Biol Reprod. 2006;75:9–16. - PubMed
-
- Simoncini T, Scorticati C, Mannella P, Fadiel A, Giretti MS, Fu XD, Baldacci C, Garibaldi S, Caruso A, Fornari L, et al. Estrogen receptor {alpha} interacts with G{alpha}13 to drive actin remodeling and endothelial cell migration via the RhoA/Rho kinase/moesin pathway. Mol Endocrinol. 2006;20:1756–1771. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
