Evolution of frequency-dependent mate choice: keeping up with fashion trends
- PMID: 17360285
- PMCID: PMC2176183
- DOI: 10.1098/rspb.2007.0043
Evolution of frequency-dependent mate choice: keeping up with fashion trends
Abstract
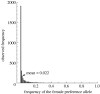
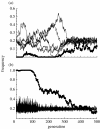
The diversity of sexual traits favoured by females is enormous and, curiously, includes preferences for males with rare or novel phenotypes. We modelled the evolution of a preference for rarity that yielded two surprising results. First, a Fisherian 'sexy son' effect can boost female preferences to a frequency well above that predicted by mutation-selection balance, even if there are significant mortality costs for females. Preferences do not reach fixation, however, as they are subject to frequency-dependent selection: if choosy females are too common, then rare genotypes in one generation become common, and thus unattractive, in the offspring generation. Nevertheless, even at relatively low frequency, preferences maintain polymorphism in male traits. The second unexpected result is that the preferences can evolve to much higher frequencies if choice is hindered, such that females cannot always express their preferences. Our results emphasize the need to consider feedback where preferences determine the dynamics of male genotypes and vice versa. They also highlight the similarity between the arbitrariness of behavioural norms in models of social evolution with punishment (the so-called 'folk theorem') and the diversity of sexual traits that can be preferred simply because deviating from the norm produces unattractive offspring and is, in this sense, 'punished'.
Figures

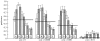
References
-
- Amos W, Worthington Wilmer J, Fullard K, Burg T.M, Croxall J.P, Bloch D, Coulson T. The influence of parental relatedness on reproductive success. Proc. R. Soc. B. 2001;268:2021–2027. doi:10.1098/rspb.2001.1751 - DOI - PMC - PubMed
-
- Björklund M. Mate choice for indirect benefits displayed by a large ornament: simulations using a neural network. Anim. Behav. 2006;71:549–553. doi:10.1016/j.anbehav.2005.05.018 - DOI
-
- Blows M.W, Hoffmann A.A. A reassessment of genetic limits to evolutionary change. Ecology. 2005;86:1371–1384.
-
- Blows M.W, Chenoweth S.F, Hine E. Orientation of the genetic variance–covariance matrix and the fitness surface for multiple male sexually selected traits. Am. Nat. 2004;163:329–340. doi:10.1086/381941 - DOI - PubMed
-
- Boyd R. Reciprocity: you have to think different. J. Evol. Biol. 2006;19:1380–1382. doi:10.1111/j.1420-9101.2006.01159.x - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
