Neuronal growth and survival mediated by eIF5A, a polyamine-modified translation initiation factor
- PMID: 17360499
- PMCID: PMC1820731
- DOI: 10.1073/pnas.0611609104
Neuronal growth and survival mediated by eIF5A, a polyamine-modified translation initiation factor
Abstract
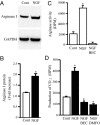
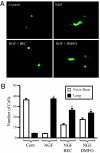
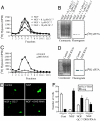
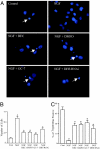
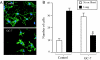
Eukaryotic translation initiation factor 5A (eIF5A), the only known protein containing the polyamine-derived amino acid hypusine, modulates protein synthesis. We show that neurotrophic and neuroprotective actions of nerve growth factor (NGF) are mediated by hypusinated eIF5A, which can account for the known roles of polyamines in cell growth and survival. NGF treatment of PC12 cells stimulates eIF5A formation. Moreover, prevention of hypusine formation by a selective inhibitor of deoxyhypusine synthase and by its depletion with RNA interference blocks the NGF-elicited augmentation of neurite outgrowth and cell survival of PC12 cells. In brain cultures, inhibition of hypusine formation also inhibits neuronal process extension.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

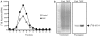
Similar articles
-
The post-translational synthesis of a polyamine-derived amino acid, hypusine, in the eukaryotic translation initiation factor 5A (eIF5A).J Biochem. 2006 Feb;139(2):161-9. doi: 10.1093/jb/mvj034. J Biochem. 2006. PMID: 16452303 Free PMC article. Review.
-
Independent roles of eIF5A and polyamines in cell proliferation.Biochem J. 2005 Feb 1;385(Pt 3):779-85. doi: 10.1042/BJ20041477. Biochem J. 2005. PMID: 15377278 Free PMC article.
-
Functional significance of eIF5A and its hypusine modification in eukaryotes.Amino Acids. 2010 Feb;38(2):491-500. doi: 10.1007/s00726-009-0408-7. Epub 2009 Dec 8. Amino Acids. 2010. PMID: 19997760 Free PMC article. Review.
-
Inactivation of eukaryotic initiation factor 5A (eIF5A) by specific acetylation of its hypusine residue by spermidine/spermine acetyltransferase 1 (SSAT1).Biochem J. 2011 Jan 1;433(1):205-13. doi: 10.1042/BJ20101322. Biochem J. 2011. PMID: 20942800 Free PMC article.
-
Production of active recombinant eIF5A: reconstitution in E.coli of eukaryotic hypusine modification of eIF5A by its coexpression with modifying enzymes.Protein Eng Des Sel. 2011 Mar;24(3):301-9. doi: 10.1093/protein/gzq110. Epub 2010 Dec 2. Protein Eng Des Sel. 2011. PMID: 21131325 Free PMC article.
Cited by
-
Insulin action on protein synthesis and its association with eIF5A expression and hypusination.Mol Biol Rep. 2019 Feb;46(1):587-596. doi: 10.1007/s11033-018-4512-1. Epub 2018 Dec 5. Mol Biol Rep. 2019. PMID: 30519811
-
Bi-allelic variants in DOHH, catalyzing the last step of hypusine biosynthesis, are associated with a neurodevelopmental disorder.Am J Hum Genet. 2022 Aug 4;109(8):1549-1558. doi: 10.1016/j.ajhg.2022.06.010. Epub 2022 Jul 19. Am J Hum Genet. 2022. PMID: 35858628 Free PMC article.
-
Manipulation of DHPS activity affects dendritic morphology and expression of synaptic proteins in primary rat cortical neurons.Front Cell Neurosci. 2024 Oct 14;18:1465011. doi: 10.3389/fncel.2024.1465011. eCollection 2024. Front Cell Neurosci. 2024. PMID: 39469305 Free PMC article.
-
Changes in arginase isoforms in a murine model of neonatal brain hypoxia-ischemia.Pediatr Res. 2021 Mar;89(4):830-837. doi: 10.1038/s41390-020-0978-3. Epub 2020 May 28. Pediatr Res. 2021. PMID: 32464635 Free PMC article.
-
uORFs with unusual translational start codons autoregulate expression of eukaryotic ornithine decarboxylase homologs.Proc Natl Acad Sci U S A. 2008 Jul 22;105(29):10079-84. doi: 10.1073/pnas.0801590105. Epub 2008 Jul 14. Proc Natl Acad Sci U S A. 2008. PMID: 18626014 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

