TGFbeta promotes conversion of CD16+ peripheral blood NK cells into CD16- NK cells with similarities to decidual NK cells
- PMID: 17360654
- PMCID: PMC1805591
- DOI: 10.1073/pnas.0611098104
TGFbeta promotes conversion of CD16+ peripheral blood NK cells into CD16- NK cells with similarities to decidual NK cells
Abstract
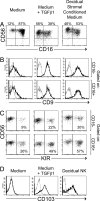
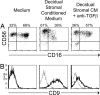
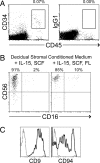
During pregnancy the uterine decidua is populated by large numbers of natural killer (NK) cells with a phenotype CD56(superbright)CD16(-)CD9(+)KIR(+) distinct from both subsets of peripheral blood NK cells. Culture of highly purified CD16(+)CD9(-) peripheral blood NK cells in medium containing TGFbeta1 resulted in a transition to CD16(-)CD9(+) NK cells resembling decidual NK cells. Decidual stromal cells, when isolated and cultured in vitro, were found to produce TGFbeta1. Incubation of peripheral blood NK cells with conditioned medium from decidual stromal cells mirrored the effects of TGFbeta1. Similar changes may occur upon NK cell entry into the decidua or other tissues expressing substantial TGFbeta. In addition, Lin(-)CD34(+)CD45(+) hematopoietic stem/progenitor cells could be isolated from decidual tissue. These progenitors also produced NK cells when cultured in conditioned medium from decidual stromal cells supplemented with IL-15 and stem cell factor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
The effect of interleukin 2 and transforming growth factor-beta 2 (TGF-beta 2) on the proliferation and natural killer activity of decidual CD16- CD56bright natural killer cells.Cell Immunol. 1993 Dec;152(2):605-13. doi: 10.1006/cimm.1993.1316. Cell Immunol. 1993. PMID: 7504983
-
Glycodelin-A stimulates the conversion of human peripheral blood CD16-CD56bright NK cell to a decidual NK cell-like phenotype.Hum Reprod. 2019 Apr 1;34(4):689-701. doi: 10.1093/humrep/dey378. Hum Reprod. 2019. PMID: 30597092
-
[The expression of chemokine receptors in CD56(bright) CD16- natural killer cells and the mechanism of their recruitment in decidua].Zhonghua Yi Xue Za Zhi. 2004 Jun 17;84(12):1018-23. Zhonghua Yi Xue Za Zhi. 2004. PMID: 15312539 Chinese.
-
Human Innate Lymphoid Cells: Their Functional and Cellular Interactions in Decidua.Front Immunol. 2018 Aug 14;9:1897. doi: 10.3389/fimmu.2018.01897. eCollection 2018. Front Immunol. 2018. PMID: 30154799 Free PMC article. Review.
-
Heterogeneity of NK Cells and Other Innate Lymphoid Cells in Human and Murine Decidua.Front Immunol. 2019 Feb 8;10:170. doi: 10.3389/fimmu.2019.00170. eCollection 2019. Front Immunol. 2019. PMID: 30800126 Free PMC article. Review.
Cited by
-
Decidual Natural Killer Cells: A Good Nanny at the Maternal-Fetal Interface During Early Pregnancy.Front Immunol. 2021 May 12;12:663660. doi: 10.3389/fimmu.2021.663660. eCollection 2021. Front Immunol. 2021. PMID: 34054831 Free PMC article. Review.
-
Renal cell tumors convert natural killer cells to a proangiogenic phenotype.Oncotarget. 2020 Jun 30;11(26):2571-2585. doi: 10.18632/oncotarget.27654. eCollection 2020 Jun 30. Oncotarget. 2020. PMID: 32655841 Free PMC article.
-
Perinatal Mesenchymal Stromal Cells and Their Possible Contribution to Fetal-Maternal Tolerance.Cells. 2019 Nov 7;8(11):1401. doi: 10.3390/cells8111401. Cells. 2019. PMID: 31703272 Free PMC article. Review.
-
Role of Macrophages in Pregnancy and Related Complications.Arch Immunol Ther Exp (Warsz). 2019 Oct;67(5):295-309. doi: 10.1007/s00005-019-00552-7. Epub 2019 Jul 8. Arch Immunol Ther Exp (Warsz). 2019. PMID: 31286151 Free PMC article. Review.
-
CD56bright natural killer (NK) cells: an important NK cell subset.Immunology. 2009 Apr;126(4):458-65. doi: 10.1111/j.1365-2567.2008.03027.x. Immunology. 2009. PMID: 19278419 Free PMC article. Review.
References
-
- Moffett-King A. Nat Rev Immunol. 2002;2:656–663. - PubMed
-
- Dunn CL, Kelly RW, Critchley HO. Reprod Biomed Online. 2003;7:151–161. - PubMed
-
- King A. Hum Reprod Update. 2000;6:28–36. - PubMed
-
- Cooper MA, Fehniger TA, Caligiuri MA. Trends Immunol. 2001;22:633–640. - PubMed
-
- Jacobs R, Hintzen G, Kemper A, Beul K, Kempf S, Behrens G, Sykora KW, Schmidt RE. Eur J Immunol. 2001;31:3121–3127. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

