Selective retention of herpes simplex virus-specific T cells in latently infected human trigeminal ganglia
- PMID: 17360672
- PMCID: PMC1805572
- DOI: 10.1073/pnas.0610847104
Selective retention of herpes simplex virus-specific T cells in latently infected human trigeminal ganglia
Abstract
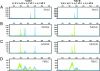
Primary infection with herpes simplex virus 1 (HSV-1) and varicella zoster virus (VZV) results in lifelong latent infections of neurons in sensory ganglia such as the trigeminal ganglia (TG). It has been postulated that T cells retained in TG inhibit reactivation of latent virus. The acquisition of TG specimens of individuals within hours after death offered the unique opportunity to characterize the phenotype and specificity of TG-resident T cells in humans. High numbers of activated CD8(+) T cells expressing a late effector memory phenotype were found to reside in latently infected TG. The T cell infiltrate was oligoclonal, and T cells selectively clustered around HSV-1 but not VZV latently infected neurons. Neuronal damage was not observed despite granzyme B expression by the neuron-interacting CD8(+) T cells. The TG-resident T cells, mainly CD8(+) T cells, were directed against HSV-1 and not to VZV, despite neuronal expression of VZV proteins. The results implicate that herpesvirus latency in human TG is associated with a local, persistent T cell response, comprising activated late effector memory CD8(+) T cells that appear to control HSV-1 latency by noncytolytic pathways. In contrast, T cells do not seem to be directly involved in controlling VZV latency in human TG.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
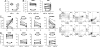

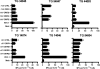
References
-
- Roizman B, Knipe DM. In: Fields Virology. Knipe DM, Howley P, editors. Philadelphia: Lippincott Williams & Wilkens; 2001. pp. 2399–2443.
-
- Cohen JI, Strauss SI. In: Fields Virology. Knipe DM, Howley P, editors. Philadelphia: Lippincott Williams & Wilkens; 2001. pp. 2707–2730.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

