Detection of anti-leishmania (Leishmania) chagasi immunoglobulin G by flow cytometry for cure assessment following chemotherapeutic treatment of American visceral leishmaniasis
- PMID: 17360851
- PMCID: PMC1865639
- DOI: 10.1128/CVI.00354-06
Detection of anti-leishmania (Leishmania) chagasi immunoglobulin G by flow cytometry for cure assessment following chemotherapeutic treatment of American visceral leishmaniasis
Abstract
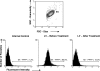
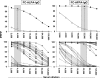
The residual serological reactivity observed in patients cured of visceral leishmaniasis (VL) represents the major factor underlying the low efficiency of most anti-Leishmania serological approaches to assess posttherapeutic cure in VL. Herein, we have described a detuned flow cytometry-based methodology to detect anti-live (FC-ALPA-immunoglobulin G [IgG]) and anti-fixed (FC-AFPA-IgG) L. chagasi promastigote IgG, along the titration curve (1:2,000 to 1:128,000), as a tool to assess late (12 months after treatment [12 mAT]) and early (2 and 6 mAT) posttherapeutic cure of pediatric American visceral leishmaniasis. Reactivities were reported as the percentage of positive fluorescent parasite (PPFP), using a PPFP of 50% as a cutoff to segregate positive and negative results. Our data demonstrated that both FC-ALPA-IgG at 1:4,000 and FC-ALPA-IgG at 1:32,000 are useful for late cure assessment in VL, with 100% specificity and outstanding likelihood ratio indices. Cure assessment at 6 mAT also showed promising performance indices, identifying 81% and 71.4% of the treated patients with negative results. However, new interpretation parameters were necessary to monitor cure at 2 mAT. We then introduced the differential PPFP (DeltaPPFP) of 25% as a new cutoff for early cure assessment at specific serum dilutions to analyze IgG reactivity by FC-ALPA-IgG and FC-AFPA-IgG. Our data demonstrated that at 2 mAT, DeltaPPFP was >25% in 60% and 57.1% of treated patients, whereas at 6 mAT, a DeltaPPFP of >25% was observed in 100% and 95.2% of samples assayed by FC-ALPA-IgG and FC-AFPA-IgG, respectively. Together, our findings showed the potential of both FC-ALPA-IgG and FC-AFPA-IgG regarding their applicability to detect differential serological reactivity and further contribution to posttherapeutic cure assessment in VL.
Figures


Similar articles
-
Anti-Leishmania chagasi immunoglobulin G3 detected by flow cytometry for early cure assessment in American visceral leishmaniasis.J Immunol Methods. 2010 Aug 31;360(1-2):76-83. doi: 10.1016/j.jim.2010.06.011. Epub 2010 Jun 17. J Immunol Methods. 2010. PMID: 20598707
-
Anti-fixed Leishmania chagasi promastigotes IgG antibodies detected by flow cytometry (FC-AFPA-IgG) as a tool for serodiagnosis and for post-therapeutic cure assessment in American visceral leishmaniasis.J Immunol Methods. 2009 Oct 31;350(1-2):36-45. doi: 10.1016/j.jim.2009.07.004. Epub 2009 Jul 25. J Immunol Methods. 2009. PMID: 19635482
-
Clinical value of anti-Leishmania (Leishmania) chagasi IgG titers detected by flow cytometry to distinguish infected from vaccinated dogs.Vet Immunol Immunopathol. 2007 Mar 15;116(1-2):85-97. doi: 10.1016/j.vetimm.2007.01.002. Epub 2007 Jan 13. Vet Immunol Immunopathol. 2007. PMID: 17287029
-
Visceral leishmaniasis in hematopoietic cell transplantation: Case report and review of the literature.J Infect Chemother. 2018 Dec;24(12):990-994. doi: 10.1016/j.jiac.2018.05.008. Epub 2018 Aug 8. J Infect Chemother. 2018. PMID: 30098915 Review.
-
Molecular targets and pathways for the treatment of visceral leishmaniasis.Drug Discov Today. 2018 Jan;23(1):161-170. doi: 10.1016/j.drudis.2017.09.006. Epub 2017 Sep 14. Drug Discov Today. 2018. PMID: 28919438 Review.
Cited by
-
Leishmaniases diagnosis: an update on the use of immunological and molecular tools.Cell Biosci. 2015 Jun 17;5:31. doi: 10.1186/s13578-015-0021-2. eCollection 2015. Cell Biosci. 2015. PMID: 26097678 Free PMC article.
-
American tegumentary leishmaniasis diagnosis using L. (V.) braziliensis fixed promastigotes: a comparative performance of serological tests and spontaneous cure identification.BMC Infect Dis. 2019 Nov 29;19(1):1015. doi: 10.1186/s12879-019-4642-7. BMC Infect Dis. 2019. PMID: 31783798 Free PMC article.
-
Biosensors for Detecting Lymphocytes and Immunoglobulins.Biosensors (Basel). 2020 Oct 27;10(11):155. doi: 10.3390/bios10110155. Biosensors (Basel). 2020. PMID: 33121071 Free PMC article. Review.
-
A Leishmania-specific hypothetical protein expressed in both promastigote and amastigote stages of Leishmania infantum employed for the serodiagnosis of, and as a vaccine candidate against, visceral leishmaniasis.Parasit Vectors. 2015 Jul 11;8:363. doi: 10.1186/s13071-015-0964-5. Parasit Vectors. 2015. PMID: 26160291 Free PMC article.
-
A New Flow Cytometry-Based Single Platform for Universal and Differential Serodiagnosis of HTLV-1/2 Infection.Front Immunol. 2022 Apr 14;13:795815. doi: 10.3389/fimmu.2022.795815. eCollection 2022. Front Immunol. 2022. PMID: 35493505 Free PMC article.
References
-
- Anam, K., F. Afrin, D. Banerjee, N. Pramanik, S. K. Guha, R. P. Goswami, S. K. Saha, and N. Ali. 1999. Differential decline in Leishmania membrane antigen-specific immunoglobulin G (IgG), IgM, IgE, and IgG subclass antibodies in Indian kala-azar patients after chemotherapy. Infect. Immun. 67:6663-6669. - PMC - PubMed
-
- Desjeux, P. 1996. Leishmaniasis—public health aspects and control. Clin. Dermatol. 14:417-423. - PubMed
-
- Galvão, L. M., R. M. Nunes, J. R. Cancado, Z. Brener, and A. U. Krettli. 1993. Lytic antibody titre as a means of assessing cure after treatment of Chagas disease: a 10 years follow-up study. Trans. R. Soc. Trop. Med. Hyg. 87:220-223. - PubMed
-
- Greiner, M., D. Pfeiffer, and R. D. Smith. 2000. Principles and practical application of the receiver-operating characteristic analysis for diagnostic tests. Prev. Vet. Med. 45:23-41. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

