Quantitative conformationally sampled pharmacophore for delta opioid ligands: reevaluation of hydrophobic moieties essential for biological activity
- PMID: 17367120
- PMCID: PMC2631547
- DOI: 10.1021/jm0612463
Quantitative conformationally sampled pharmacophore for delta opioid ligands: reevaluation of hydrophobic moieties essential for biological activity
Abstract
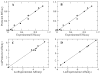
Recent studies have indicated several therapeutic applications for delta opioid agonists and antagonists. To exploit the therapeutic potential of delta opioids developing a structural basis for the activity of ligands at the delta opioid receptor is essential. The conformationally sampled pharmacophore (CSP) method (Bernard et al. J. Am. Chem. Soc. 2003, 125, 3103-3107) is extended here to obtain quantitative models of delta opioid ligand efficacy and affinity. Quantification is performed via overlap integrals of the conformational space sampled by ligands with respect to a reference compound. Iterative refinement of the CSP model identified hydrophobic groups other than the traditional phenylalanine residues as important for efficacy and affinity in DSLET and ICI 174 864. The obtained models for a structurally diverse set of peptidic and nonpeptidic delta opioid ligands offer good predictions with R2 values>0.9, and the predicted efficacy for a set of test compounds was consistent with the experimental values.
Figures



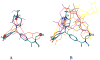
Similar articles
-
Conformationally sampled pharmacophore for peptidic delta opioid ligands.J Med Chem. 2005 Dec 1;48(24):7773-80. doi: 10.1021/jm050785p. J Med Chem. 2005. PMID: 16302816
-
2D conformationally sampled pharmacophore: a ligand-based pharmacophore to differentiate delta opioid agonists from antagonists.J Am Chem Soc. 2003 Mar 12;125(10):3101-7. doi: 10.1021/ja027644m. J Am Chem Soc. 2003. PMID: 12617677
-
De novo design, synthesis, and biological activities of high-affinity and selective non-peptide agonists of the delta-opioid receptor.J Med Chem. 1998 Nov 19;41(24):4767-76. doi: 10.1021/jm980374r. J Med Chem. 1998. PMID: 9822547
-
δ Opioid Receptor Inverse Agonists and their In Vivo Pharmacological Effects.Curr Top Med Chem. 2020;20(31):2889-2902. doi: 10.2174/1568026620666200402115654. Curr Top Med Chem. 2020. PMID: 32238139 Review.
-
Nonpeptidic delta (delta) opioid agonists and antagonists of the diarylmethylpiperazine class: what have we learned?Top Curr Chem. 2011;299:121-40. doi: 10.1007/128_2010_83. Top Curr Chem. 2011. PMID: 21630509 Review.
Cited by
-
Molecular switch controlling the binding of anionic bile acid conjugates to human apical sodium-dependent bile acid transporter.J Med Chem. 2010 Jun 24;53(12):4749-60. doi: 10.1021/jm1003683. J Med Chem. 2010. PMID: 20504026 Free PMC article.
-
Consensus 3D model of μ-opioid receptor ligand efficacy based on a quantitative Conformationally Sampled Pharmacophore.J Phys Chem B. 2011 Jun 9;115(22):7487-96. doi: 10.1021/jp202542g. Epub 2011 May 12. J Phys Chem B. 2011. PMID: 21563754 Free PMC article.
-
Synthesis, modeling, and pharmacological evaluation of UMB 425, a mixed μ agonist/δ antagonist opioid analgesic with reduced tolerance liabilities.ACS Chem Neurosci. 2013 Sep 18;4(9):1256-66. doi: 10.1021/cn4000428. Epub 2013 Jun 11. ACS Chem Neurosci. 2013. PMID: 23713721 Free PMC article.
-
Characterizing structural transitions using localized free energy landscape analysis.PLoS One. 2009;4(5):e5525. doi: 10.1371/journal.pone.0005525. Epub 2009 May 13. PLoS One. 2009. PMID: 19436759 Free PMC article.
-
Deconstructing 14-phenylpropyloxymetopon: minimal requirements for binding to mu opioid receptors.Bioorg Med Chem. 2012 Jul 15;20(14):4556-63. doi: 10.1016/j.bmc.2012.05.006. Epub 2012 May 11. Bioorg Med Chem. 2012. PMID: 22677527 Free PMC article.
References
-
- Martin TJ, Eisenach JC. Pharmacology of Opioid and Nonopioid Analgesics in Chronic Pain States. J Pharmacol Exp Ther. 2001;299:811–817. - PubMed
-
- Coop A, MacKerell AD., Jr The future of opioid analgesics. Am J Pharm Educ. 2003;66:153–156.
-
- White JM, Irvine RJ. Mechanisms of Fatal Opioid Overdose. Addiction. 1999;94:961–972. - PubMed
-
- Pappagallo M. Incidence, prevalence and management of opioid bowel dysfunction. Am J Surg. 2001;182:S11–S18. - PubMed
-
- Chakrabarti S, Wang L, Tang WJ, Gintzler AR. Chronic Morphine Augments Adenylyl Cyclase Phosphorylation: Relevance to Altered Signaling during Tolerance/Dependence. Mol Pharmacol. 1998;54:949–953. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

