Donor natural killer cell allorecognition of missing self in haploidentical hematopoietic transplantation for acute myeloid leukemia: challenging its predictive value
- PMID: 17371948
- PMCID: PMC1896125
- DOI: 10.1182/blood-2006-07-038687
Donor natural killer cell allorecognition of missing self in haploidentical hematopoietic transplantation for acute myeloid leukemia: challenging its predictive value
Abstract
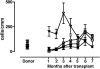
We analyzed 112 patients with high-risk acute myeloid leukemia (61 in complete remission [CR]; 51 in relapse), who received human leukocyte-antigen (HLA)-haploidentical transplants from natural killer (NK) alloreactive (n = 51) or non-NK alloreactive donors (n = 61). NK alloreactive donors possessed HLA class I, killer-cell immunoglobulin-like receptor (KIR) ligand(s) which were missing in the recipients, KIR gene(s) for missing self recognition on recipient targets, and alloreactive NK clones against recipient targets. Transplantation from NK-alloreactive donors was associated with a significantly lower relapse rate in patients transplanted in CR (3% versus 47%) (P > .003), better event-free survival in patients transplanted in relapse (34% versus 6%, P = .04) and in remission (67% versus 18%, P = .02), and reduced risk of relapse or death (relative risk versus non-NK-alloreactive donor, 0.48; 95% CI, 0.29-0.78; P > .001). In all patients we tested the "missing ligand" model which pools KIR ligand mismatched transplants and KIR ligand-matched transplants from donors possessing KIR(s) for which neither donor nor recipient have HLA ligand(s). Only transplantation from NK-alloreactive donors is associated with a survival advantage.
Figures

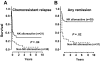


References
-
- Aversa F, Tabilio A, Terenzi A, et al. Successful engraftment of T-cell-depleted haploidentical “three-loci” incompatible transplants in leukemia patients by addition of recombinant human granulocyte colony-stimulating factor-mobilized peripheral blood progenitor cells to bone marrow inoculum. Blood. 1994;84:3948–3955. - PubMed
-
- Aversa F, Tabilio A, Velardi A, et al. Treatment of high-risk acute leukemia with T-cell-depleted stem cells from related donors with one fully mismatched HLA haplotype. N Engl J Med. 1998;339:1186–1193. - PubMed
-
- Aversa F, Terenzi A, Tabilio A, et al. Full haplotype-mismatched hematopoietic stem-cell transplantation: a phase II study in patients with acute leukemia at high risk of relapse. J Clin Oncol. 2005;23:3447–3454. - PubMed
-
- Ruggeri L, Capanni M, Urbani E, et al. Effectiveness of donor natural killer cell alloreactivity in mismatched hematopoietic transplants. Science. 2002;295:2097–2100. - PubMed
-
- Karre K, Ljunggren HG, Piontek G, Kiessling R. Selective rejection of H-2-deficient lymphoma variants suggests alternative immune defence strategy. Nature. 1986;319:675–678. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

