Cytokine-mediated disruption of lymphocyte trafficking, hemopoiesis, and induction of lymphopenia, anemia, and thrombocytopenia in anti-CD137-treated mice
- PMID: 17371976
- PMCID: PMC2770095
- DOI: 10.4049/jimmunol.178.7.4194
Cytokine-mediated disruption of lymphocyte trafficking, hemopoiesis, and induction of lymphopenia, anemia, and thrombocytopenia in anti-CD137-treated mice
Abstract
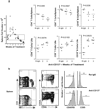
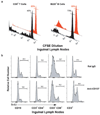
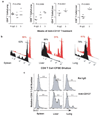
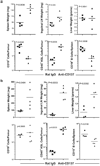
CD137-mediated signals costimulate T cells and protect them from activation-induced apoptosis; they induce curative antitumor immunity and enhance antiviral immune responses in mice. In contrast, anti-CD137 agonistic mAbs can suppress T-dependent humoral immunity and reverse the course of established autoimmune disease. These results have provided a rationale for assessing the therapeutic potential of CD137 ligands in human clinical trials. In this study, we report that a single 200-mug injection of anti-CD137 given to otherwise naive BALB/c or C57BL/6 mice led to the development of a series of immunological anomalies. These included splenomegaly, lymphadenopathy, hepatomegaly, multifocal hepatitis, anemia, altered trafficking of B cells and CD8 T cells, loss of NK cells, and a 10-fold increase in bone marrow (BM) cells bearing the phenotype of hemopoietic stem cells. These events were dependent on CD8 T cells, TNF-alpha, IFN-gamma, and type I IFNs. BM cells up-regulated Fas, and there was a significant increase in the number of CD8+ T cells that correlated with a loss of CD19+ and Ab-secreting cells in the BM. TCR Valphabeta usage was random and polyclonal among liver-infiltrating CD8 T cells, and multifocal CD8+ T cell infiltrates were resolved upon termination of anti-CD137 treatment. Anti-CD137-treated mice developed lymphopenia, thrombocytopenia, and anemia, and had lowered levels of hemoglobin and increased numbers of reticulocytes.
Conflict of interest statement
The authors have no financial conflict of interest.
Figures












Similar articles
-
Therapeutic antitumor efficacy of anti-CD137 agonistic monoclonal antibody in mouse models of myeloma.Clin Cancer Res. 2008 Nov 1;14(21):6895-906. doi: 10.1158/1078-0432.CCR-08-0285. Clin Cancer Res. 2008. PMID: 18980984 Free PMC article.
-
Treatment with anti-CD137 mAbs causes intense accumulations of liver T cells without selective antitumor immunotherapeutic effects in this organ.Cancer Immunol Immunother. 2010 Aug;59(8):1223-33. doi: 10.1007/s00262-010-0846-9. Epub 2010 Mar 25. Cancer Immunol Immunother. 2010. PMID: 20336294 Free PMC article.
-
Anti-CD137 antibodies in the treatment of autoimmune disease and cancer.Immunol Res. 2004;29(1-3):197-208. doi: 10.1385/IR:29:1-3:197. Immunol Res. 2004. PMID: 15181282 Review.
-
Combined TIM-3 blockade and CD137 activation affords the long-term protection in a murine model of ovarian cancer.J Transl Med. 2013 Sep 17;11:215. doi: 10.1186/1479-5876-11-215. J Transl Med. 2013. PMID: 24044888 Free PMC article.
-
[The immunotherapy potential of agonistic anti-CD137 (4-1BB) monoclonal antibodies for malignancies and chronic viral diseases].An Sist Sanit Navar. 2006 Jan-Apr;29(1):77-96. doi: 10.4321/s1137-66272006000100007. An Sist Sanit Navar. 2006. PMID: 16670731 Review. Spanish.
Cited by
-
Therapeutic antitumor efficacy of anti-CD137 agonistic monoclonal antibody in mouse models of myeloma.Clin Cancer Res. 2008 Nov 1;14(21):6895-906. doi: 10.1158/1078-0432.CCR-08-0285. Clin Cancer Res. 2008. PMID: 18980984 Free PMC article.
-
A novel form of 4-1BBL has better immunomodulatory activity than an agonistic anti-4-1BB Ab without Ab-associated severe toxicity.Vaccine. 2009 Dec 11;28(2):512-22. doi: 10.1016/j.vaccine.2009.09.127. Epub 2009 Oct 29. Vaccine. 2009. PMID: 19836479 Free PMC article.
-
SA-4-1BBL as the immunomodulatory component of a HPV-16 E7 protein based vaccine shows robust therapeutic efficacy in a mouse cervical cancer model.Vaccine. 2010 Aug 16;28(36):5794-802. doi: 10.1016/j.vaccine.2010.06.073. Epub 2010 Jul 4. Vaccine. 2010. PMID: 20603135 Free PMC article.
-
Boosting Cancer Immunotherapy with Anti-CD137 Antibody Therapy.Clin Cancer Res. 2015 Jul 15;21(14):3113-20. doi: 10.1158/1078-0432.CCR-15-0263. Epub 2015 Apr 23. Clin Cancer Res. 2015. PMID: 25908780 Free PMC article. Review.
-
Evaluating the cellular targets of anti-4-1BB agonist antibody during immunotherapy of a pre-established tumor in mice.PLoS One. 2010 Jun 8;5(6):e11003. doi: 10.1371/journal.pone.0011003. PLoS One. 2010. PMID: 20543982 Free PMC article.
References
-
- Watts TH. TNF/TNFR family members in costimulation of T cell responses. Annu. Rev. Immunol. 2005;23:23–68. - PubMed
-
- Melero I, Shuford WW, Newby SA, Aruffo A, Ledbetter JA, Hellstrom KE, Mittler RS, Chen L. Monoclonal antibodies against the 4-1BB T-cell activation molecule eradicate established tumors. Nat. Med. 1997;3:682–685. - PubMed
-
- Melero I, Bach N, Hellstrom KE, Aruffo A, Mittler RS, Chen L. Amplification of tumor immunity by gene transfer of the co-stimulatory 4-1BB ligand: synergy with the CD28 costimulatory pathway. Eur. J. Immunol. 1998;28:1116–1121. - PubMed
-
- Wang J, Guo Z, Dong Y, Kim O, Hart J, Adams A, Larsen CP, Mittler RS, Newell KA. Role of 4-1BB in allograft rejection mediated by CD8+ T cells. Am. J. Transplant. 2003;3:543–551. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

