Genome-wide coorientation of replication and transcription reduces adverse effects on replication in Bacillus subtilis
- PMID: 17372224
- PMCID: PMC1838449
- DOI: 10.1073/pnas.0608999104
Genome-wide coorientation of replication and transcription reduces adverse effects on replication in Bacillus subtilis
Abstract
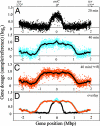
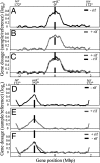
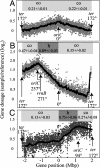
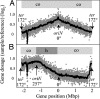
In many bacteria, there is a strong bias for genes to be encoded on the leading strand of DNA, resulting in coorientation of replication and transcription. In Bacillus subtilis, transcription of the majority of genes (75%) is cooriented with replication. By using genome-wide profiling of replication with DNA microarrays, we found that this coorientation bias reduces adverse effects of transcription on replication. We found that in wild-type cells, transcription did not appear to affect the rate of replication elongation. However, in mutants with reversed transcription bias for an extended region of the chromosome, replication elongation was slower. This reduced replication rate depended on transcription and was limited to the region in which the directions of replication and transcription are opposed. These results support the hypothesis that the strong bias to coorient transcription and replication is due to selective pressure for processive, efficient, and accurate replication.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

