Microfluidic immunoassays as rapid saliva-based clinical diagnostics
- PMID: 17374724
- PMCID: PMC1828942
- DOI: 10.1073/pnas.0607254104
Microfluidic immunoassays as rapid saliva-based clinical diagnostics
Abstract
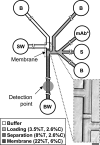
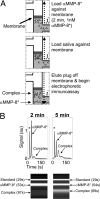
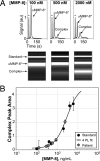
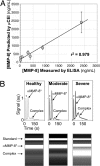
At present, point-of-care (POC) diagnostics typically provide a binary indication of health status (e.g., home pregnancy test strip). Before anticipatory use of diagnostics for assessment of complex diseases becomes widespread, development of sophisticated bioassays capable of quantitatively measuring disease biomarkers is necessary. Successful translation of new bioassays into clinical settings demands the ability to monitor both the onset and progression of disease. Here we report on a clinical POC diagnostic that enables rapid quantitation of an oral disease biomarker in human saliva by using a monolithic disposable cartridge designed to operate in a compact analytical instrument. Our microfluidic method facilitates hands-free saliva analysis by integrating sample pretreatment (filtering, enrichment, mixing) with electrophoretic immunoassays to quickly measure analyte concentrations in minimally pretreated saliva samples. Using 20 microl of saliva, we demonstrate rapid (<10 min) measurement of the collagen-cleaving enzyme matrix metalloproteinase-8 (MMP-8) in saliva from healthy and periodontally diseased subjects. In addition to physiologically measurable indicators of periodontal disease, conventional measurements of salivary MMP-8 were used to validate the microfluidic assays described in this proof-of-principle study. The microchip-based POC diagnostic demonstrated is applicable to rapid, reliable measurement of proteinaceous disease biomarkers in biological fluids.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Kricka LJ. Clin Chem. 1998;44:2008–2014. - PubMed
-
- Schulte TH, Bardell RL, Weigl BH. Clin Chim Acta. 2002;321:1–10. - PubMed
-
- Christodoulides N, Mohanty S, Miller CS, Langub MC, Floriano PN, Dharshan P, Ali MF, Bernard B, Romanovicz D, Anslyn E, Fox PC, McDevitt JT. Lab Chip. 2005;5:261–269. - PubMed
-
- Yang CY, Brooks E, Li Y, Denny P, Ho CM, Qi FX, Shi WY, Wolinsky L, Wu B, Wong DTW, Montemagno CD. Lab Chip. 2005;5:1017–1023. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

