A positive regulatory loop between foxi3a and foxi3b is essential for specification and differentiation of zebrafish epidermal ionocytes
- PMID: 17375188
- PMCID: PMC1810426
- DOI: 10.1371/journal.pone.0000302
A positive regulatory loop between foxi3a and foxi3b is essential for specification and differentiation of zebrafish epidermal ionocytes
Abstract
Background: Epidermal ionocytes play essential roles in the transepithelial transportation of ions, water, and acid-base balance in fish embryos before their branchial counterparts are fully functional. However, the mechanism controlling epidermal ionocyte specification and differentiation remains unknown.
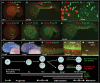
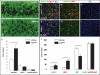
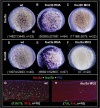
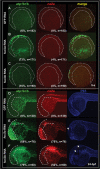
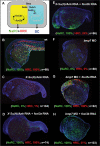
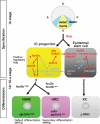
Methodology/principal findings: In zebrafish, we demonstrated that Delta-Notch-mediated lateral inhibition plays a vital role in singling out epidermal ionocyte progenitors from epidermal stem cells. The entire epidermal ionocyte domain of genetic mutants and morphants, which failed to transmit the DeltaC-Notch1a/Notch3 signal from sending cells (epidermal ionocytes) to receiving cells (epidermal stem cells), differentiates into epidermal ionocytes. The low Notch activity in epidermal ionocyte progenitors is permissive for activating winged helix/forkhead box transcription factors of foxi3a and foxi3b. Through gain- and loss-of-function assays, we show that the foxi3a-foxi3b regulatory loop functions as a master regulator to mediate a dual role of specifying epidermal ionocyte progenitors as well as of subsequently promoting differentiation of Na(+),K(+)-ATPase-rich cells and H(+)-ATPase-rich cells in a concentration-dependent manner.
Conclusions/significance: This study provides a framework to show the molecular mechanism controlling epidermal ionocyte specification and differentiation in a low vertebrate for the first time. We propose that the positive regulatory loop between foxi3a and foxi3b not only drives early ionocyte differentiation but also prevents the complete blockage of ionocyte differentiation when the master regulator of foxi3 function is unilaterally compromised.
Conflict of interest statement
Figures



References
-
- Wall SM. Recent advances in our understanding of intercalated cells. Curr Opin Nephrol Hypertens. 2005;14:480–484. - PubMed
-
- Hulander M, Kiernan AE, Blomqvist SR, Carlsson P, Samuelsson EJ, et al. Lack of pendrin expression leads to deafness and expansion of the endolymphatic compartment in inner ears of Foxi1 null mutant mice. Development. 2003;130:2013–2025. - PubMed
-
- Perry SF, Shahsavarani A, Georgalis T, Bayaa M, Furimsky M, et al. Channels, pumps, and exchangers in the gill and kidney of freshwater fishes: Their role in ionic and acid-base regulation. J Exp Zoolog A Comp Exp Biol. 2003;300:53–62. - PubMed
-
- Evans DH, Piermarini PM, Choe KP. The multifunctional fish gill: dominant site of gas exchange, osmoregulation, acid-base regulation, and excretion of nitrogenous waste. Physiol Rev. 2005;85:97–177. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

