Ionotropic glutamate receptor GluR1 in the visual cortex of hamster: distribution and co-localization with calcium-binding proteins and GABA
- PMID: 17375209
- PMCID: PMC1828082
- DOI: 10.1267/ahc.05058
Ionotropic glutamate receptor GluR1 in the visual cortex of hamster: distribution and co-localization with calcium-binding proteins and GABA
Abstract
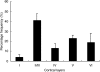
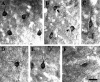
The subunit composition of the AMPA receptor is critical to its function. AMPA receptors that display very low calcium permeability include the GluR2 subunit, while AMPA receptors that contain other subunits, such as GluR1, display high calcium permeability. We have studied the distribution and morphology of neurons containing GluR1 in the hamster visual cortex with antibody immunocytochemistry. We compared this labeling to that for calbindin D28K, parvalbumin, and GABA. Anti-GluR1-immunoreactive (IR) neurons were located in all layers. The highest density of GluR1-IR neurons was found in layers II/III. The labeled neurons were non-pyramidal neurons, but were varied in morphology. The majority of the labeled neurons were round or oval cells. However, stellate, vertical fusiform, pyriform, and horizontal neurons were also labeled with the anti-GluR1 antibody. Two-color immunofluorescence revealed that many of the GluR1-IR neurons in the hamster visual cortex were double-labeled with either calbindin D28K (31.50%), or parvalbumin (22.91%), or GABA (63.89%). These results indicate that neurons in the hamster visual cortex express GluR1 differently according to different layers and selective cell types, and that many of the GluR1-IR neurons are limited to neurons that express calbindin D28K, parvalbumin, or GABA. The present study elucidates the neurochemical structure of GluR1, a useful clue in understanding the differential vulnerability of GluR1-containing neurons with regard to calcium-dependent excitotoxic mechanisms.
Figures


Similar articles
-
Distribution of AMPA glutamate receptor GluR1 subunit-immunoreactive neurons and their co-localization with calcium-binding proteins and GABA in the mouse visual cortex.Mol Cells. 2006 Feb 28;21(1):34-41. Mol Cells. 2006. PMID: 16511345
-
Ionotropic glutamate receptor GluR2/3-immunoreactive neurons in the cat, rabbit, and hamster superficial superior colliculus.Neurosci Res. 2004 Jun;49(2):139-55. doi: 10.1016/j.neures.2004.02.009. Neurosci Res. 2004. PMID: 15140557
-
Glutamate receptors GluR1 and GluR4 in the hamster superior colliculus: distribution and co-localization with calcium-binding proteins and GABA.Acta Histochem Cytochem. 2009 Apr 28;42(2):29-38. doi: 10.1267/ahc.08035. Epub 2009 Mar 31. Acta Histochem Cytochem. 2009. PMID: 19492025 Free PMC article.
-
Immunocytochemical localization of calcium-binding proteins, calbindin D28K-, calretinin-, and parvalbumin-containing neurons in the dog visual cortex.Zoolog Sci. 2011 Sep;28(9):694-702. doi: 10.2108/zsj.28.694. Zoolog Sci. 2011. PMID: 21882959
-
The organization of GABAergic neurons in the mammalian superior colliculus.Prog Brain Res. 1992;90:219-48. doi: 10.1016/s0079-6123(08)63616-x. Prog Brain Res. 1992. PMID: 1321459 Review.
Cited by
-
Genome-wide gene expression profiling in GluR1 knockout mice: key role of the calcium signaling pathway in glutamatergically mediated hippocampal transmission.Eur J Neurosci. 2009 Dec;30(12):2318-26. doi: 10.1111/j.1460-9568.2009.07022.x. Epub 2009 Dec 10. Eur J Neurosci. 2009. PMID: 20092574 Free PMC article.
-
Calcyon upregulation in adolescence impairs response inhibition and working memory in adulthood.Mol Psychiatry. 2011 Jun;16(6):672-84. doi: 10.1038/mp.2011.14. Epub 2011 Mar 15. Mol Psychiatry. 2011. PMID: 21403673 Free PMC article.
References
-
- Baimbridge K. G., Celio M. R., Rogers J. H. Calcium-binding proteins in the nervous system. Trends Neurosci. 1992;15:303–308. - PubMed
-
- Blumcke I., Hof P. R., Morrison J. H., Celio M. R. Distribution of parvalbumin immunoreactivity in the visual cortex of Old World monkeys and humans. J. Comp. Neurol. 1990;301:417–432. - PubMed
-
- Celio M. R. Calbindin D-28k and parvalbumin in the rat nervous system. Neuroscience. 1990;35:375–475. - PubMed
-
- Cellerino A., Siciliano R., Domenici L., Maffei L. Parvalbumin immunoreactivity: a reliable marker for the effects of monocular deprivation in the rat visual cortex. Neuroscience. 1992;51:749–753. - PubMed

