Kv2 subunits underlie slowly inactivating potassium current in rat neocortical pyramidal neurons
- PMID: 17379638
- PMCID: PMC2170822
- DOI: 10.1113/jphysiol.2007.128454
Kv2 subunits underlie slowly inactivating potassium current in rat neocortical pyramidal neurons
Abstract
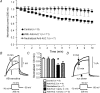
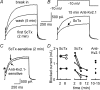
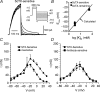
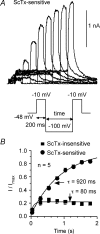
We determined the expression of Kv2 channel subunits in rat somatosensory and motor cortex and tested for the contributions of Kv2 subunits to slowly inactivating K+ currents in supragranular pyramidal neurons. Single cell RT-PCR showed that virtually all pyramidal cells expressed Kv2.1 mRNA and approximately 80% expressed Kv2.2 mRNA. Immunocytochemistry revealed striking differences in the distribution of Kv2.1 and Kv2.2 subunits. Kv2.1 subunits were clustered and located on somata and proximal dendrites of all pyramidal cells. Kv2.2 subunits were primarily distributed on large apical dendrites of a subset of pyramidal cells from deep layers. We used two methods for isolating currents through Kv2 channels after excluding contributions from Kv1 subunits: intracellular diffusion of Kv2.1 antibodies through the recording pipette and extracellular application of rStromatoxin-1 (ScTx). The Kv2.1 antibody specifically blocked the slowly inactivating K+ current by 25-50% (at 8 min), demonstrating that Kv2.1 subunits underlie much of this current in neocortical pyramidal neurons. ScTx (300 nM) also inhibited approximately 40% of the slowly inactivating K+ current. We observed occlusion between the actions of Kv2.1 antibody and ScTx. In addition, Kv2.1 antibody- and ScTx-sensitive currents demonstrated similar recovery from inactivation and voltage dependence and kinetics of activation and inactivation. These data indicate that both agents targeted the same channels. Considering the localization of Kv2.1 and 2.2 subunits, currents from truncated dissociated cells are probably dominated by Kv2.1 subunits. Compared with Kv2.1 currents in expression systems, the Kv2.1 current in neocortical pyramidal cells activated and inactivated at relatively negative potentials and was very sensitive to holding potential.
Figures







Similar articles
-
Postnatal development of A-type and Kv1- and Kv2-mediated potassium channel currents in neocortical pyramidal neurons.J Neurophysiol. 2011 Jun;105(6):2976-88. doi: 10.1152/jn.00758.2010. Epub 2011 Mar 30. J Neurophysiol. 2011. PMID: 21451062 Free PMC article.
-
Comparison of the endogenous IK currents in rat hippocampal neurons and cloned Kv2.1 channels in CHO cells.Cell Biol Int. 2008 Dec;32(12):1514-20. doi: 10.1016/j.cellbi.2008.08.019. Epub 2008 Aug 30. Cell Biol Int. 2008. PMID: 18801450
-
Expression and biophysical properties of Kv1 channels in supragranular neocortical pyramidal neurones.J Physiol. 2006 Mar 1;571(Pt 2):371-89. doi: 10.1113/jphysiol.2005.097006. Epub 2005 Dec 22. J Physiol. 2006. PMID: 16373387 Free PMC article.
-
Kv2.1 and silent Kv subunits underlie the delayed rectifier K+ current in cultured small mouse DRG neurons.Am J Physiol Cell Physiol. 2009 Jun;296(6):C1271-8. doi: 10.1152/ajpcell.00088.2009. Epub 2009 Apr 8. Am J Physiol Cell Physiol. 2009. PMID: 19357235 Free PMC article.
-
The contribution of Kv2.2-mediated currents decreases during the postnatal development of mouse dorsal root ganglion neurons.Physiol Rep. 2016 Mar;4(6):e12731. doi: 10.14814/phy2.12731. Epub 2016 Mar 31. Physiol Rep. 2016. PMID: 27033450 Free PMC article.
Cited by
-
Sodium and potassium conductances in principal neurons of the mouse piriform cortex: a quantitative description.J Physiol. 2018 Nov;596(22):5397-5414. doi: 10.1113/JP275824. Epub 2018 Oct 14. J Physiol. 2018. PMID: 30194865 Free PMC article.
-
Chemokine co-receptor CCR5/CXCR4-dependent modulation of Kv2.1 channel confers acute neuroprotection to HIV-1 glycoprotein gp120 exposure.PLoS One. 2013 Sep 24;8(9):e76698. doi: 10.1371/journal.pone.0076698. eCollection 2013. PLoS One. 2013. PMID: 24086760 Free PMC article.
-
Kv2.2: a novel molecular target to study the role of basal forebrain GABAergic neurons in the sleep-wake cycle.Sleep. 2013 Dec 1;36(12):1839-48. doi: 10.5665/sleep.3212. Sleep. 2013. PMID: 24293758 Free PMC article.
-
The Shab family potassium channels are highly enriched at the presynaptic terminals of human neurons.J Biol Chem. 2025 Mar;301(3):108235. doi: 10.1016/j.jbc.2025.108235. Epub 2025 Jan 27. J Biol Chem. 2025. PMID: 39880095 Free PMC article.
-
Formation of heteromeric Kv2 channels in mammalian brain neurons.J Biol Chem. 2010 May 14;285(20):15048-15055. doi: 10.1074/jbc.M109.074260. Epub 2010 Mar 4. J Biol Chem. 2010. PMID: 20202934 Free PMC article.
References
-
- Antonucci DE, Lim ST, Vassanelli S, Trimmer JS. Dynamic localization and clustering of dendritic Kv2.1 voltage-dependent potassium channels in developing hippocampal neurons. Neuroscience. 2001;108:69–81. - PubMed
-
- Archer SL, Souil E, Dinh-Xuan AT, Schremmer B, Mercier JC, El Yaagoubi A, Nguyen-Huu L, Reeve HL, Hampl V. Molecular identification of the role of voltage-gated K+ channels, Kv1.5 and Kv2.1, in hypoxic pulmonary vasoconstriction and control of resting membrane potential in rat pulmonary myocytes. J Clin Invest. 1998;101:2319–2330. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

