Desmin stimulates differentiation of cardiomyocytes and up-regulation of brachyury and nkx2.5
- PMID: 17381547
- PMCID: PMC7615841
- DOI: 10.1111/j.1432-0436.2007.00162.x
Desmin stimulates differentiation of cardiomyocytes and up-regulation of brachyury and nkx2.5
Abstract
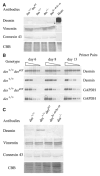
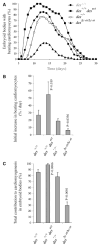
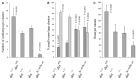
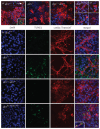
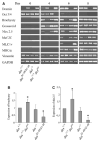
Desmin contributes to structural integrity and function of the myocardium but its function seems to be redundant in early cardiomyogenesis in the desmin null mouse model. To test the hypothesis that desmin also plays a supportive role in cardiomyogenic commitment and early differentiation of cardiomyocytes we investigated cardiomyogenesis in embryoid bodies expressing different desmin alleles. Constitutive expression of desmin and increased synthesis during mesoderm formation led to the up-regulation of brachyury and nkx2.5 genes, accelerated early cardiomyogenesis and resulted in the development of large, proliferating, highly interconnected, and synchronously beating cardiomyocyte clusters, whereas desmin null cardiomyocytes featured an opposite phenotype. In contrast, constitutive expression of amino-terminally truncated desmin(Delta1-48) interfered with the beginning of cardiomyogenesis, caused down-regulation of mesodermal and myocardial transcription factors, and hampered myofibrillogenesis and survival of cardiomyocytes. These results provide first evidence that a type III intermediate filament protein takes part in regulating the differentiation of mesoderm to cardiomyocytes at the very beginning of cardiomyogenesis.
Figures

Similar articles
-
Differentiation of cardiomyocytes requires functional serine residues within the amino-terminal domain of desmin.Differentiation. 2007 Sep;75(7):616-26. doi: 10.1111/j.1432-0436.2007.00163.x. Epub 2007 Mar 23. Differentiation. 2007. PMID: 17381546 Free PMC article.
-
Myocyte enhancer factor 2C and Nkx2-5 up-regulate each other's expression and initiate cardiomyogenesis in P19 cells.J Biol Chem. 1998 Dec 25;273(52):34904-10. doi: 10.1074/jbc.273.52.34904. J Biol Chem. 1998. PMID: 9857019
-
The cellular prion protein identifies bipotential cardiomyogenic progenitors.Circ Res. 2010 Jan 8;106(1):111-9. doi: 10.1161/CIRCRESAHA.109.209478. Epub 2009 Nov 12. Circ Res. 2010. PMID: 19910576
-
Fbxo25 controls Tbx5 and Nkx2-5 transcriptional activity to regulate cardiomyocyte development.Biochim Biophys Acta. 2015 Jun;1849(6):709-21. doi: 10.1016/j.bbagrm.2015.02.002. Epub 2015 Feb 25. Biochim Biophys Acta. 2015. PMID: 25725482
-
Maturation of human induced pluripotent stem cell-derived cardiomyocytes promoted by Brachyury priming.Sci Rep. 2025 Apr 24;15(1):14399. doi: 10.1038/s41598-025-97676-y. Sci Rep. 2025. PMID: 40275010 Free PMC article.
Cited by
-
Desmin related disease: a matter of cell survival failure.Curr Opin Cell Biol. 2015 Feb;32:113-20. doi: 10.1016/j.ceb.2015.01.004. Epub 2015 Feb 11. Curr Opin Cell Biol. 2015. PMID: 25680090 Free PMC article. Review.
-
Numb family proteins play roles in Desmin and Vimentin localization at the Z-disc.J Muscle Res Cell Motil. 2025 Mar;46(1):9-22. doi: 10.1007/s10974-024-09687-3. Epub 2024 Dec 15. J Muscle Res Cell Motil. 2025. PMID: 39674848
-
Embryonic stem cells facilitate the isolation of persistent clonal cardiovascular progenitor cell lines and leukemia inhibitor factor maintains their self-renewal and myocardial differentiation potential in vitro.Cells Tissues Organs. 2013;197(4):249-68. doi: 10.1159/000345804. Epub 2013 Jan 22. Cells Tissues Organs. 2013. PMID: 23343517 Free PMC article.
-
Intermediate filaments in cardiomyopathy.Biophys Rev. 2018 Aug;10(4):1007-1031. doi: 10.1007/s12551-018-0443-2. Epub 2018 Jul 19. Biophys Rev. 2018. PMID: 30027462 Free PMC article. Review.
-
Effects of increased preload on the force-frequency response and contractile kinetics in early stages of cardiac muscle hypertrophy.Am J Physiol Heart Circ Physiol. 2012 Jun 15;302(12):H2509-17. doi: 10.1152/ajpheart.00660.2011. Epub 2012 Mar 30. Am J Physiol Heart Circ Physiol. 2012. PMID: 22467304 Free PMC article.
References
-
- Bader A, Al-Dubai H, Weitzer G. Leukemia inhibitory factor modulates cardiogenesis in embryoid bodies in opposite fashions. Circ Res. 2000;86:787–794. - PubMed
-
- Balogh J, Li Z, Paulin D, Arner A. Lower active force generation and improved fatigue resistance in skeletal muscle from desmin deficient mice. J Muscle Res Cell Motil. 2003;24:453–459. - PubMed
-
- Balogh J, Merisckay M, Li Z, Paulin D, Arner A. Hearts from mice lacking desmin have a myopathy with impaired active force generation and unaltered wall compliance. Cardiovasc Res. 2002;53:439–450. - PubMed
-
- Capetanaki C. Desmin cytoskeleton in healthy and failing heart. Heart Fail Rev. 2000;5:203–220. - PubMed
-
- Capetanaki Y. Desmin cytoskeleton: a potential regulator of muscle mitochondrial behavior and function. Trends Cardiovasc Med. 2002;12:339–348. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

