Aldose reductase mediates endotoxin-induced production of nitric oxide and cytotoxicity in murine macrophages
- PMID: 17382209
- PMCID: PMC1885210
- DOI: 10.1016/j.freeradbiomed.2007.01.033
Aldose reductase mediates endotoxin-induced production of nitric oxide and cytotoxicity in murine macrophages
Abstract
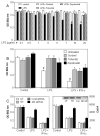
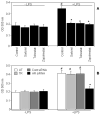
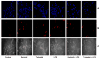
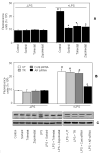
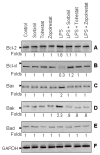
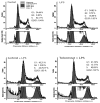
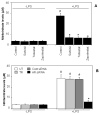
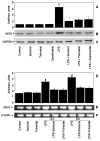
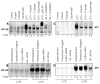
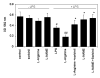
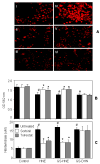
Aldose reductase (AR) is a ubiquitously expressed protein with pleiotrophic roles as an efficient catalyst for the reduction of toxic lipid aldehydes and mediator of hyperglycemia, cytokine, and growth factor-induced redox-sensitive signals that cause secondary diabetic complications. Although AR inhibition has been shown to be protective against oxidative stress signals, the role of AR in regulating nitric oxide (NO) synthesis and NO-mediated apoptosis has not been elucidated to date. We therefore investigated the role of AR in regulating lipopolysaccharide (LPS)-induced NO synthesis and apoptosis in RAW 264.7 macrophages. Inhibition or RNA interference ablation of AR suppressed LPS-stimulated production of NO and overexpression of iNOS mRNA. Inhibition or ablation of AR also prevented the LPS-induced apoptosis, cell cycle arrest, activation of caspase-3, p38-MAPK, JNK, NF-kappaB, and AP1. In addition, AR inhibition prevented the LPS-induced down-regulation of Bcl-xl and up-regulation of Bax and Bak in macrophages. L-Arginine increased and L-NAME decreased the severity of cell death caused by LPS and AR inhibitors prevented it. Furthermore, inhibition of AR prevents cell death caused by HNE and GS-HNE, but not GS-DHN. Our findings for the first time suggest that AR-catalyzed lipid aldehyde-glutathione conjugates regulate the LPS-induced production of inflammatory marker NO and cytotoxicity in RAW 264.7 cells. Inhibition or ablation of AR activity may be a potential therapeutic target in endotoximia and other inflammatory diseases.
Figures

References
-
- Raetz CR, Ulevitch RJ, Wright SD, Sibley CH, Ding A, Nathan CF. Gram-negative endotoxin: an extraordinary lipid with profound effects on eukaryotic signal transduction. FASEB J. 1991;5:2652–2660. - PubMed
-
- Heine H, Rietschel ET, Ulmer AJ. The biology of endotoxin. Mol Biotechnol. 2001;19:279–296. - PubMed
-
- Lopez-Bojorquez LN, Dehesa AZ, Reyes-Teran G. Molecular mechanisms involved in the pathogenesis of septic shock. Arch Med Res. 2004;35:465–479. - PubMed
-
- Wolkow PP. Involvement and dual effects of nitric oxide in septic shock. Inflamm Res. 1998;47:52–166. - PubMed
-
- Baumgarten G, Knuefermann P, Schuhmacher G, Vervolgyi V, von Rappard J, Dreiner U, Fink K, Djoufack C, Hoeft A, Grohe C, Knowlton AA, Meyer R. Toll-like receptor 4, nitric oxide, and myocardial depression in endotoxemia. Shock. 2006;25:43–49. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

