Adaptive TGF-beta-dependent regulatory T cells control autoimmune diabetes and are a privileged target of anti-CD3 antibody treatment
- PMID: 17389382
- PMCID: PMC1851030
- DOI: 10.1073/pnas.0701171104
Adaptive TGF-beta-dependent regulatory T cells control autoimmune diabetes and are a privileged target of anti-CD3 antibody treatment
Abstract
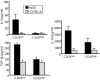
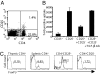
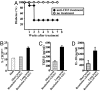
Previous results have shown that CD4(+)CD25(+) regulatory T cells (Tregs) control autoimmunity in a spontaneous model of type 1 diabetes, the nonobese diabetic (NOD) mouse. Moreover, anti-CD3 reverses diabetes in this setting by promoting Tregs that function in a TGF-beta-dependent manner. This finding contrasts with a large body of work suggesting that CD4(+)CD25(high) Tregs act in a cytokine-independent manner, thus suggesting that another type of Treg is operational in this setting. We sought to determine the basis of suppression both in untreated NOD mice and in those treated with anti-CD3. Our present results show that a subset of foxP3(+) cells present within a CD4(+)CD25(low) lymphocyte subset suppresses T cell immunity in spontaneously diabetic NOD mice in a TGF-beta-dependent manner, a functional property typical of "adaptive" regulatory T cells. This distinct Treg subset is evident in NOD, but not normal, mice, suggesting that the NOD mice may generate these adaptive Tregs in an attempt to regulate ongoing autoimmunity. Importantly, in two distinct in vivo models, these TGF-beta-dependent adaptive CD4(+)CD25(low) T cells can be induced from peripheral CD4(+)CD25(-) T lymphocytes by anti-CD3 immunotherapy which correlates with the restoration of self-tolerance.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
-
- Sakaguchi S, Ono M, Setoguchi R, Yagi H, Hori S, Fehervari Z, Shimizu J, Takahashi T, Nomura T. Immunol Rev. 2006;212:8–27. - PubMed
-
- Roncarolo MG, Gregori S, Battaglia M, Bacchetta R, Fleischhauer K, Levings MK. Immunol Rev. 2006;212:28–50. - PubMed
-
- Shevach EM, DiPaolo RA, Andersson J, Zhao DM, Stephens GL, Thornton AM. Immunol Rev. 2006;212:60–73. - PubMed
-
- Cobbold SP, Adams E, Graca L, Daley S, Yates S, Paterson A, Robertson NJ, Nolan KF, Fairchild PJ, Waldmann H. Immunol Rev. 2006;213:239–255. - PubMed
-
- Umetsu DT, DeKruyff RH. Immunol Rev. 2006;212:238–255. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

