Coordinate expression and functional profiling identify an extracellular proteolytic signaling pathway
- PMID: 17389401
- PMCID: PMC1838401
- DOI: 10.1073/pnas.0606514104
Coordinate expression and functional profiling identify an extracellular proteolytic signaling pathway
Abstract
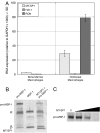
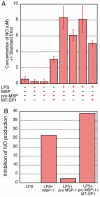
A multidisciplinary method combining transcriptional data, specificity profiling, and biological characterization of an enzyme may be used to predict novel substrates. By integrating protease substrate profiling with microarray gene coexpression data from nearly 2,000 human normal and cancerous tissue samples, three fundamental components of a protease-activated signaling pathway were identified. We find that MT-SP1 mediates extracellular signaling by regulating the local activation of the prometastatic growth factor MSP-1. We demonstrate MT-SP1 expression in peritoneal macrophages, and biochemical methods confirm the ability of MT-SP1 to cleave and activate pro-MSP-1 in vitro and in a cellular context. MT-SP1 induced the ability of MSP-1 to inhibit nitric oxide production in bone marrow macrophages. Addition of HAI-1 or an MT-SP1-specific antibody inhibitor blocked the proteolytic activation of MSP-1 at the cell surface of peritoneal macrophages. Taken together, our work indicates that MT-SP1 is sufficient for MSP-1 activation and that MT-SP1, MSP-1, and the previously shown MSP-1 tyrosine kinase receptor RON are required for peritoneal macrophage activation. This work shows that this triad of growth factor, growth factor activator protease, and growth factor receptor is a protease-activated signaling pathway. Individually, MT-SP1 and RON overexpression have been implicated in cancer progression and metastasis. Transcriptional coexpression of these genes suggests that this signaling pathway may be involved in several human cancers.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Cao J, Zheng S, Zheng L, Cai X, Zhang Y, Geng L, Fang Y. Chin Med J (English) 2001;114:726–730. - PubMed
-
- Galkin AV, Mullen L, Fox WD, Brown J, Duncan D, Moreno O, Madison EL, Agus DB. Prostate. 2004;61:228–235. - PubMed
-
- Hoang CD, D'Cunha J, Kratzke MG, Casmey CE, Frizelle SP, Maddaus MA, Kratzke RA. Chest. 2004;125:1843–1852. - PubMed
-
- Johnson MD, Oberst MD, Lin CY, Dickson RB. Exp Rev Mol Diagn. 2003;3:331–338. - PubMed
-
- Kang JY, Dolled-Filhart M, Ocal IT, Singh B, Lin CY, Dickson RB, Rimm DL, Camp RL. Cancer Res. 2003;63:1101–1105. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

