Gangliosides GM1 and GM3 in the living cell membrane form clusters susceptible to cholesterol depletion and chilling
- PMID: 17392511
- PMCID: PMC1877094
- DOI: 10.1091/mbc.e07-01-0071
Gangliosides GM1 and GM3 in the living cell membrane form clusters susceptible to cholesterol depletion and chilling
Abstract
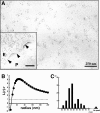
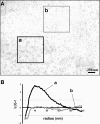
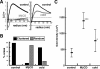
Presence of microdomains has been postulated in the cell membrane, but two-dimensional distribution of lipid molecules has been difficult to determine in the submicrometer scale. In the present paper, we examined the distribution of gangliosides GM1 and GM3, putative raft molecules in the cell membrane, by immunoelectron microscopy using quick-frozen and freeze-fractured specimens. This method physically immobilized molecules in situ and thus minimized the possibility of artifactual perturbation. By point pattern analysis of immunogold labeling, GM1 was shown to make clusters of <100 nm in diameter in normal mouse fibroblasts. GM1-null fibroblasts were not labeled, but developed a similar clustered pattern when GM1 was administered. On cholesterol depletion or chilling, the clustering of both endogenous and exogenously-loaded GM1 decreased significantly, but the distribution showed marked regional heterogeneity in the cells. GM3 also showed cholesterol-dependent clustering, and although clusters of GM1 and GM3 were found to occasionally coincide, these aggregates were separated in most cases, suggesting the presence of heterogeneous microdomains. The present method enabled to capture the molecular distribution of lipids in the cell membrane, and demonstrated that GM1 and GM3 form clusters that are susceptible to cholesterol depletion and chilling.
Figures



Comment in
-
An MBoC favorite: gangliosides GM1 and GM3 in the living cell membrane form clusters susceptible to cholesterol depletion and chilling.Mol Biol Cell. 2012 Oct;23(20):3925. doi: 10.1091/mbc.E12-02-0159. Mol Biol Cell. 2012. PMID: 23066041 Free PMC article. No abstract available.
References
-
- Brugger B., Graham C., Leibrecht I., Mombelli E., Jen A., Wieland F., Morris R. The membrane domains occupied by glycosylphosphatidylinositol-anchored prion protein and Thy-1 differ in lipid composition. J. Biol. Chem. 2004;279:7530–7536. - PubMed
-
- Chandler D. E. Comparison of quick-frozen and chemically fixed sea-urchin eggs: structural evidence that cortical granule exocytosis is preceded by a local increase in membrane mobility. J. Cell Sci. 1984;72:23–36. - PubMed
-
- Chen X., Resh M. D. Cholesterol depletion from the plasma membrane triggers ligand-independent activation of the epidermal growth factor receptor. J. Biol. Chem. 2002;277:49631–49637. - PubMed
-
- Chigorno V., Palestini P., Sciannamblo M., Dolo V., Pavan A., Tettamanti G., Sonnino S. Evidence that ganglioside enriched domains are distinct from caveolae in MDCK II and human fibroblast cells in culture. Eur. J. Biochem. 2000;267:4187–4197. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

