Bacterial infection of fly ovaries reduces egg production and induces local hemocyte activation
- PMID: 17400292
- PMCID: PMC3109252
- DOI: 10.1016/j.dci.2007.02.003
Bacterial infection of fly ovaries reduces egg production and induces local hemocyte activation
Abstract
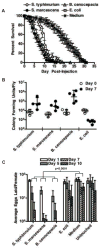
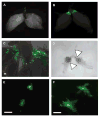
Morbidity, the state of being diseased, is an important aspect of pathogenesis that has gone relatively unstudied in fruit flies. Our interest is in characterizing how bacterial pathogenesis affects various physiologies of the fly. We chose to examine the fly ovary because we found bacterial infection had a striking effect on fly reproduction. We observed decreased egg laying after bacterial infection that correlated with increased bacterial virulence. We also found that bacteria colonized the ovary in a previously undescribed manner; bacteria were found in the posterior of the ovary, adjacent to the lateral oviduct. This local infection in the ovary resulted in melanization and activation of the cellular immune response at the site of infection.
Figures



References
-
- Lemaitre B, Nicolas E, Michaut L, Reichhart JM, Hoffmann JA. The dorsoventral regulatory gene cassette spatzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell. 1996;86(6):973–83. - PubMed
-
- Ayres JS, Schneider DS. Genomic dissection of microbial pathogenesis in cultured Drosophila cells. Trends Microbiol. 2006;14(3):101–4. - PubMed
-
- Brennan CA, Anderson KV. Drosophila: the genetics of innate immune recognition and response. Annu Rev Immunol. 2004;22:457–83. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

