RIAM links the ADAP/SKAP-55 signaling module to Rap1, facilitating T-cell-receptor-mediated integrin activation
- PMID: 17403904
- PMCID: PMC1900018
- DOI: 10.1128/MCB.02011-06
RIAM links the ADAP/SKAP-55 signaling module to Rap1, facilitating T-cell-receptor-mediated integrin activation
Abstract
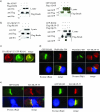
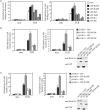
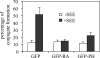
One outcome of T-cell receptor (TCR) signaling is increased affinity and avidity of integrins for their ligands. This occurs through a process known as inside-out signaling, which has been shown to require several molecular components including the adapter proteins ADAP (adhesion and degranulation-promoting adapter protein) and SKAP-55 (55-kDa src kinase-associated phosphoprotein) and the small GTPase Rap1. Herein, we provide evidence linking ADAP and SKAP-55 to RIAM, a recently described adapter protein that binds selectively to active Rap1. We identified RIAM as a key component linking the ADAP/SKAP-55 module to the small GTPase Rap1, facilitating TCR-mediated integrin activation. We show that RIAM constitutively interacts with SKAP-55 in both a heterologous transfection system and primary T cells and map the region essential for this interaction. Additionally, we find that the SKAP-55/RIAM complex is essential both for TCR-mediated adhesion and for efficient conjugate formation between T cells and antigen-presenting cells. Mechanistic studies revealed that the ADAP/SKAP-55 module relocalized RIAM and Rap1 to the plasma membrane following TCR activation to facilitate integrin activation. These results describe for the first time a link between ADAP/SKAP-55 and the Rap1/RIAM complex and provide a potential new mechanism for TCR-mediated integrin activation.
Figures

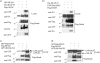


Similar articles
-
HPK1 competes with ADAP for SLP-76 binding and via Rap1 negatively affects T-cell adhesion.Eur J Immunol. 2010 Nov;40(11):3220-5. doi: 10.1002/eji.201040313. Eur J Immunol. 2010. PMID: 20957749
-
Src homology 2-domain containing leukocyte-specific phosphoprotein of 76 kDa is mandatory for TCR-mediated inside-out signaling, but dispensable for CXCR4-mediated LFA-1 activation, adhesion, and migration of T cells.J Immunol. 2009 Nov 1;183(9):5756-67. doi: 10.4049/jimmunol.0900649. Epub 2009 Oct 7. J Immunol. 2009. PMID: 19812192
-
Regulation of T-cell antigen receptor-mediated inside-out signaling by cytosolic adapter proteins and Rap1 effector molecules.Immunol Rev. 2007 Aug;218:82-91. doi: 10.1111/j.1600-065X.2007.00543.x. Immunol Rev. 2007. PMID: 17624945 Review.
-
The ADAP/SKAP55 signaling module regulates T-cell receptor-mediated integrin activation through plasma membrane targeting of Rap1.Mol Cell Biol. 2006 Oct;26(19):7130-44. doi: 10.1128/MCB.00331-06. Mol Cell Biol. 2006. PMID: 16980616 Free PMC article.
-
Small GTPases and LFA-1 reciprocally modulate adhesion and signaling.Immunol Rev. 2007 Aug;218:114-25. doi: 10.1111/j.1600-065X.2007.00538.x. Immunol Rev. 2007. PMID: 17624948 Review.
Cited by
-
ADAP and SKAP55 deficiency suppresses PD-1 expression in CD8+ cytotoxic T lymphocytes for enhanced anti-tumor immunotherapy.EMBO Mol Med. 2015 Jun;7(6):754-69. doi: 10.15252/emmm.201404578. EMBO Mol Med. 2015. PMID: 25851535 Free PMC article.
-
Concerning immune synapses: a spatiotemporal timeline.F1000Res. 2016 Mar 31;5:F1000 Faculty Rev-418. doi: 10.12688/f1000research.7796.1. eCollection 2016. F1000Res. 2016. PMID: 27092248 Free PMC article. Review.
-
The adapter protein SLP-76 mediates "outside-in" integrin signaling and function in T cells.Mol Cell Biol. 2009 Oct;29(20):5578-89. doi: 10.1128/MCB.00283-09. Epub 2009 Aug 10. Mol Cell Biol. 2009. PMID: 19667077 Free PMC article.
-
Rap1-GTP-interacting adaptor molecule (RIAM) protein controls invasion and growth of melanoma cells.J Biol Chem. 2011 May 27;286(21):18492-504. doi: 10.1074/jbc.M110.189811. Epub 2011 Mar 26. J Biol Chem. 2011. PMID: 21454517 Free PMC article.
-
The cellular context of T cell signaling.Immunity. 2009 Apr 17;30(4):482-92. doi: 10.1016/j.immuni.2009.03.010. Immunity. 2009. PMID: 19371714 Free PMC article. Review.
References
-
- Bazzoni, G., and M. E. Hemler. 1998. Are changes in integrin affinity and conformation overemphasized? Trends Biochem. Sci. 23:30-34. - PubMed
-
- Boerth, N. J., B. A. Judd, and G. A. Koretzky. 2000. Functional association between SLAP-130 and SLP-76 in Jurkat T cells. J. Biol. Chem. 275:5143-51452. - PubMed
-
- Bos, J. L. 2005. Linking Rap to cell adhesion. Curr. Opin. Cell Biol. 17:123-128. - PubMed
-
- Bos, J. L., B. Franke, L. M'Rabet, K. Reedquist, and F. Zwartkruis. 1997. In search of a function for the Ras-like GTPase Rap1. FEBS Lett. 410:59-62. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
