Prenatal exposure to elevated NT3 disrupts synaptic selectivity in the spinal cord
- PMID: 17409232
- PMCID: PMC2562665
- DOI: 10.1523/JNEUROSCI.0197-07.2007
Prenatal exposure to elevated NT3 disrupts synaptic selectivity in the spinal cord
Abstract
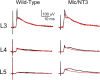
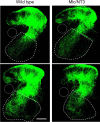
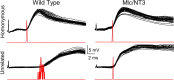
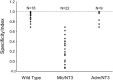
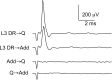
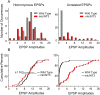
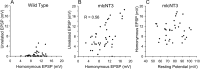
Monosynaptic connections between muscle spindle (Ia) afferents and motoneurons (MNs), the central portion of the stretch reflex circuit, are highly specific, but the mechanisms underlying this specificity are primarily unknown. In this study, we report that embryonic overexpression of neurotrophin-3 (NT3) in muscles disrupts the development of these specific Ia-MN connections, using transgenic (mlc/NT3) mice that express elevated levels of NT3 in muscles during development. In mlc/NT3 mice, there is a substantial increase in the amplitudes of monosynaptic EPSPs evoked by Ia afferents in MNs as measured with extracellular recordings from ventral roots. Despite this increased functional projection of Ia afferents, there is no obvious change in the anatomical density of Ia projections into the ventral horn of the spinal cord. Intracellular recordings from MNs revealed a major disruption in the pattern of Ia-MN connections. In addition to the normal connections between Ia afferents and MNs supplying the same muscle, there were also strong monosynaptic inputs from Ia afferents supplying unrelated muscles, which explains the increase seen in extracellular recordings. There was also a large variability in the strength of Ia input to individual MNs, both from correct and incorrect Ia afferents. Postnatal muscular administration of NT3 did not cause these changes in connectivity. These results indicate that prenatal exposure to elevated levels of NT3 disrupts the normal mechanisms responsible for synaptic selectivity in the stretch reflex circuit.
Figures


References
-
- Aldskogius H, Molander C, Persson J, Thomander L. Specific and nonspecific regeneration of motor axons after sciatic nerve injury and repair in the rat. J Neurol Sci. 1987;80:249–257. - PubMed
-
- Arber S, Ladle DR, Lin JH, Frank E, Jessell TM. ETS gene Er81 controls the formation of functional connections between group Ia sensory afferents and motor neurons. Cell. 2000;101:485–498. - PubMed
-
- Arvanian VL, Bowers WJ, Anderson A, Horner PJ, Federoff HJ, Mendell LM. Combined delivery of neurotrophin-3 and NMDA receptors 2D subunit strengthens synaptic transmission in contused and staggered double hemisected spinal cord of neonatal rat. Exp Neurol. 2006;197:347–352. - PubMed
-
- Arvanov VL, Seebach BS, Mendell LM. NT-3 evokes an LTP-like facilitation of AMPA/kainate receptor-mediated synaptic transmission in the neonatal rat spinal cord. J Neurophysiol. 2000;84:752–758. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
