PLA2 and PI3K/PTEN pathways act in parallel to mediate chemotaxis
- PMID: 17419997
- PMCID: PMC1986835
- DOI: 10.1016/j.devcel.2007.03.005
PLA2 and PI3K/PTEN pathways act in parallel to mediate chemotaxis
Abstract
Directed cell migration involves signaling events that lead to local accumulation of PI(3,4,5)P(3), but additional pathways act in parallel. A genetic screen in Dictyostelium discoideum to identify redundant pathways revealed a gene with homology to patatin-like phospholipase A(2). Loss of this gene did not alter PI(3,4,5)P(3) regulation, but chemotaxis became sensitive to reductions in PI3K activity. Likewise, cells deficient in PI3K activity were more sensitive to inhibition of PLA(2) activity. Deletion of the PLA(2) homolog and two PI3Ks caused a strong defect in chemotaxis and a reduction in receptor-mediated actin polymerization. In wild-type cells, chemoattractants stimulated a rapid burst in an arachidonic acid derivative. This response was absent in cells lacking the PLA(2) homolog, and exogenous arachidonic acid reduced their dependence on PI3K signaling. We propose that PLA(2) and PI3K signaling act in concert to mediate chemotaxis, and metabolites of PLA(2) may be important mediators of the response.
Figures
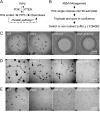


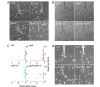
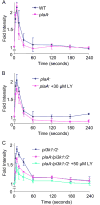
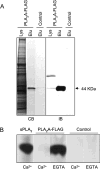
Similar articles
-
Essential role of PI3-kinase and phospholipase A2 in Dictyostelium discoideum chemotaxis.J Cell Biol. 2007 Jun 4;177(5):809-16. doi: 10.1083/jcb.200701134. Epub 2007 May 29. J Cell Biol. 2007. PMID: 17535967 Free PMC article.
-
The regulation of cell motility and chemotaxis by phospholipid signaling.J Cell Sci. 2008 Mar 1;121(Pt 5):551-9. doi: 10.1242/jcs.023333. J Cell Sci. 2008. PMID: 18287584 Free PMC article. Review.
-
Phospholipase C regulation of phosphatidylinositol 3,4,5-trisphosphate-mediated chemotaxis.Mol Biol Cell. 2007 Dec;18(12):4772-9. doi: 10.1091/mbc.e07-05-0407. Epub 2007 Sep 26. Mol Biol Cell. 2007. PMID: 17898079 Free PMC article.
-
Chemotaxis in the absence of PIP3 gradients.Curr Biol. 2007 May 1;17(9):813-7. doi: 10.1016/j.cub.2007.04.004. Curr Biol. 2007. PMID: 17462897
-
PI 3-kinases and PTEN: how opposites chemoattract.Cell. 2002 May 31;109(5):541-4. doi: 10.1016/s0092-8674(02)00765-1. Cell. 2002. PMID: 12062096 Review.
Cited by
-
Sex-Based Differences in Human Neutrophil Chemorepulsion.J Immunol. 2022 Jul 15;209(2):354-367. doi: 10.4049/jimmunol.2101103. Epub 2022 Jul 6. J Immunol. 2022. PMID: 35793910 Free PMC article.
-
Signal-activated phospholipase regulation of leukocyte chemotaxis.J Lipid Res. 2009 Apr;50 Suppl(Suppl):S231-6. doi: 10.1194/jlr.R800096-JLR200. Epub 2008 Dec 23. J Lipid Res. 2009. PMID: 19109234 Free PMC article. Review.
-
C2GAP2 is a common regulator of Ras signaling for chemotaxis, phagocytosis, and macropinocytosis.Front Immunol. 2022 Nov 29;13:1075386. doi: 10.3389/fimmu.2022.1075386. eCollection 2022. Front Immunol. 2022. PMID: 36524124 Free PMC article.
-
Small-molecule screen identifies reactive oxygen species as key regulators of neutrophil chemotaxis.Proc Natl Acad Sci U S A. 2010 Feb 23;107(8):3546-51. doi: 10.1073/pnas.0914351107. Epub 2010 Feb 8. Proc Natl Acad Sci U S A. 2010. PMID: 20142487 Free PMC article.
-
Using biomaterials to modulate chemotactic signaling for central nervous system repair.Biomed Mater. 2018 Apr 27;13(4):044106. doi: 10.1088/1748-605X/aaad82. Biomed Mater. 2018. PMID: 29411713 Free PMC article. Review.
References
-
- Ackermann EJ, Kempner ES, Dennis EA. Ca2+-independent cytosolic phospholipase A2 from Macrophage-like P388D1 cells. J Biol Chem. 1994;269:9227–9233. - PubMed
-
- Ackermann EJ, Conde-Frieboes K, Dennis EA. Inhibition of macrophage Ca2+-independent phospholipase A2 by bromoenol lactone and trifluoromethyl ketones. J Biol Chem. 1995;270:445–450. - PubMed
-
- Adachi H, Hasebe T, Yoshinaga K, Ohta T, Sutoh K. Isolation of Dictyostelium discoideum cytokinesis mutants by restriction enzyme-mediated integration of the Blasticidin S resistance marker. Biochem Biophys Res Commun. 1994;205:1808–1814. - PubMed
-
- Baggiolini M. Chemokines in pathology and medicine. J Intern Med. 2001;250:91–104. - PubMed
-
- Buczynski G, Grove B, Nomura A, Kleve M, Bush J, Firtel RA, Cardelli J. Inactivation of two Dictyostelium discoideum genes, DdPIK1 and DdPIK2, encoding proteins related to mammalian phosphatidylinositide 3-kinases, results in defects in endocytosis, lysosome to postlysosome transport, and actin cytoskeleton organization. J Cell Biol. 1997;136:1271–1286. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

